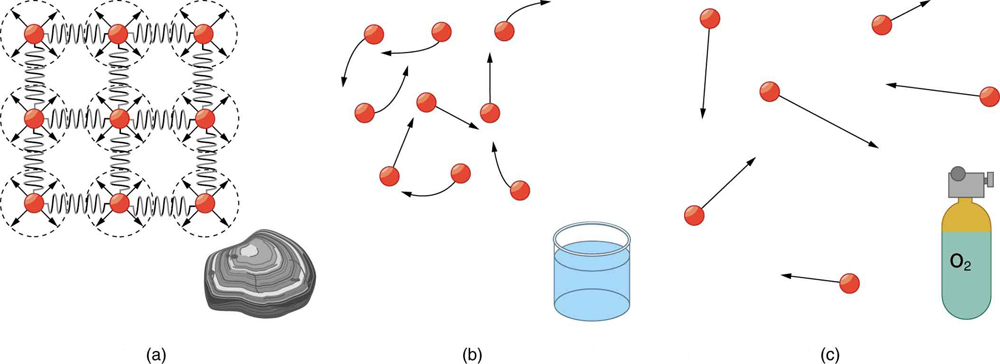
Matter most commonly exists as a solid, liquid, or gas; these states are known as the three common phases of matter. Solids have a definite shape and a specific volume, liquids have a definite volume but their shape changes depending on the container in which they are held, and gases have neither a definite shape nor a specific volume as their molecules move to fill the container in which they are held. (See [Figure 1].) Liquids and gases are considered to be fluids because they yield to shearing forces, whereas solids resist them. Note that the extent to which fluids yield to shearing forces (and hence flow easily and quickly) depends on a quantity called the viscosity which is discussed in detail in Viscosity and Laminar Flow; Poiseuille’s Law. We can understand the phases of matter and what constitutes a fluid by considering the forces between atoms that make up matter in the three phases.

Atoms in solids are in close contact, with forces between them that allow the atoms to vibrate but not to change positions with neighboring atoms. (These forces can be thought of as springs that can be stretched or compressed, but not easily broken.) Thus, a solid resists all types of stress. A solid cannot be easily deformed because the atoms that make up the solid are not able to move about freely. Solids also resist compression, because their atoms form part of a lattice structure in which the atoms are a relatively fixed distance apart. Under compression, the atoms would be forced into one another. Most of the examples we have studied so far have involved solid objects which deform very little when stressed.
Atomic and molecular characteristics explain and underlie the macroscopic characteristics of solids and fluids. This submicroscopic explanation is one theme of this text and is highlighted in the Things Great and Small features in Conservation of Momentum. See, for example, microscopic description of collisions and momentum or microscopic description of pressure in a gas. This present section is devoted entirely to the submicroscopic explanation of solids and liquids.
In contrast, liquids deform easily when stressed and do not spring back to their original shape once the force is removed because the atoms are free to slide about and change neighbors—that is, they flow (so they are a type of fluid), with the molecules held together by their mutual attraction. When a liquid is placed in a container with no lid on, it remains in the container ( providing the container has no holes below the surface of the liquid!). Because the atoms are closely packed, liquids, like solids, resist compression.
Atoms in gases are separated by distances that are large compared with the size of the atoms. The forces between gas atoms are therefore very weak, except when the atoms collide with one another. Gases thus not only flow (and are therefore considered to be fluids) but they are relatively easy to compress because there is much space and little force between atoms. When placed in an open container gases, unlike liquids, will escape. The major distinction is that gases are easily compressed, whereas liquids are not. We shall generally refer to both gases and liquids simply as fluids, and make a distinction between them only when they behave differently.
Heat, cool, and compress atoms and molecules and watch as they change between solid, liquid, and gas phases.
What physical characteristic distinguishes a fluid from a solid?
Strategy: To distinguish fluids from solids, we must consider how each state of matter responds to applied forces, specifically shearing (sideways) forces.
Solution: The key physical characteristic that distinguishes a fluid from a solid is that a fluid yields to and flows under shearing forces, while a solid resists and maintains its shape.
At the molecular level, this difference arises from the bonding between atoms:
Discussion: This distinction explains everyday observations: a block of ice maintains its shape under its own weight, but water takes the shape of its container. Similarly, honey flows slowly when tilted because it yields to shearing forces (though its high viscosity means it flows slowly). The ability to flow is the defining characteristic of fluids, which is why both liquids and gases are classified as fluids despite their other differences.
Which of the following substances are fluids at room temperature: air, mercury, water, glass?
Strategy: To identify fluids, we apply the defining characteristic: fluids are substances that yield to shearing forces and flow. We evaluate each substance at room temperature (approximately 20°C to 25°C).
Solution: At room temperature, air, mercury, and water are fluids, while glass is a solid.
Analysis of each substance:
Discussion: A common misconception is that glass is a “very slow-flowing liquid” based on observations of old window panes being thicker at the bottom. However, this thickness variation is due to historical manufacturing processes, not flow. Glass at room temperature is many orders of magnitude more viscous than any practical fluid—it would take longer than the age of the universe for glass to noticeably flow at room temperature. Therefore, glass is properly classified as a solid.
Why are gases easier to compress than liquids and solids?
Strategy: To understand compressibility differences, we must examine the molecular structure and spacing in each phase of matter.
Solution: Gases are easier to compress than liquids and solids because gas molecules are separated by large distances compared to their size, with mostly empty space between them.
The molecular explanation:
Discussion: This explains practical applications: air in a bicycle tire compresses easily when pumped, allowing more air molecules to fit in the same volume. Water in a hydraulic system, however, transmits pressure almost instantaneously because it barely compresses—this incompressibility is essential for hydraulic brakes and lifts. At standard temperature and pressure, air is about 20,000 times more compressible than water. The compressibility of gases also explains why scuba tanks can hold large volumes of breathing gas and why compressed natural gas vehicles store fuel at high pressures.
How do gases differ from liquids?
Strategy: Both gases and liquids are fluids (they flow under shearing forces), so we need to identify the key properties that distinguish them from each other.
Solution: While both gases and liquids are fluids, they differ in several important ways:
| Property | Liquids | Gases |
|---|---|---|
| Compressibility | Nearly incompressible | Highly compressible |
| Molecular spacing | Molecules nearly in contact | Molecules widely separated |
| Definite volume | Yes—maintains volume | No—expands to fill container |
| Intermolecular forces | Strong attractive forces | Weak forces (except during collisions) |
| Density | High (similar to solids) | Low (typically 1000× less than liquids) |
| Container requirement | Stays in open container | Must be in closed container |
Discussion: These differences arise from molecular behavior:
The high compressibility of gases versus the near-incompressibility of liquids is the most practically important distinction, enabling applications like pneumatic tools (which use compressible air) versus hydraulic systems (which use incompressible liquids for precise force transmission).