Physical characteristics that are quantized—such as energy, charge, and angular momentum—are of such importance that names and symbols are given to them. The values of quantized entities are expressed in terms of quantum numbers, and the rules governing them are of the utmost importance in determining what nature is and does. This section covers some of the more important quantum numbers and rules—all of which apply in chemistry, material science, and far beyond the realm of atomic physics, where they were first discovered. Once again, we see how physics makes discoveries which enable other fields to grow.
The energy states of bound systems are quantized, because the particle wavelength can fit into the bounds of the system in only certain ways. This was elaborated for the hydrogen atom, for which the allowed energies are expressed as ${E}_{n}\propto 1/{n}^{2}$ , where $n=1, 2, 3, ...$ . We define $n$ to be the principal quantum number that labels the basic states of a system. The lowest-energy state has $n=1$ , the first excited state has $n=2$ , and so on. Thus the allowed values for the principal quantum number are
This is more than just a numbering scheme, since the energy of the system, such as the hydrogen atom, can be expressed as some function of $n$ , as can other characteristics (such as the orbital radii of the hydrogen atom).
The fact that the magnitude of angular momentum is quantized was first recognized by Bohr in relation to the hydrogen atom; it is now known to be true in general. With the development of quantum mechanics, it was found that the magnitude of angular momentum $L$ can have only the values
where $l$ is defined to be the angular momentum quantum number. The rule for $l$ in atoms is given in the parentheses. Given $n$ , the value of $l$ can be any integer from zero up to $n-1$ . For example, if $n=4$ , then $l$ can be 0, 1, 2, or 3.
Note that for $n=1$ , $l$ can only be zero. This means that the ground-state angular momentum for hydrogen is actually zero, not $h/2\pi$ as Bohr proposed. The picture of circular orbits is not valid, because there would be angular momentum for any circular orbit. A more valid picture is the cloud of probability shown for the ground state of hydrogen in [Figure 3]. The electron actually spends time in and near the nucleus. The reason the electron does not remain in the nucleus is related to Heisenberg’s uncertainty principle—the electron’s energy would have to be much too large to be confined to the small space of the nucleus. Now the first excited state of hydrogen has $n=2$ , so that $l$ can be either 0 or 1, according to the rule in $L=\sqrt{l\left(l+1\right)}\frac{h}{2\pi }$ . Similarly, for $n=3$ , $l$ can be 0, 1, or 2. It is often most convenient to state the value of $l$ , a simple integer, rather than calculating the value of $L$ from $L=\sqrt{l\left(l+1\right)}\frac{h}{2\pi }$ . For example, for $l=2$ , we see that
It is much simpler to state $l=2$.
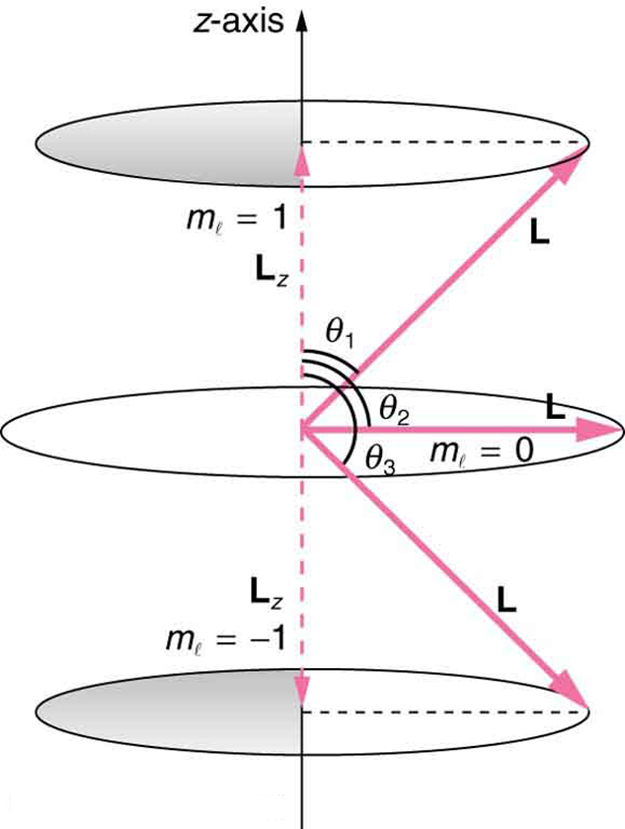
As recognized in the Zeeman effect, the direction of angular momentum is quantized. We now know this is true in all circumstances. It is found that the component of angular momentum along one direction in space, usually called the $z$ -axis, can have only certain values of ${L}_{z}$ . The direction in space must be related to something physical, such as the direction of the magnetic field at that location. This is an aspect of relativity. Direction has no meaning if there is nothing that varies with direction, as does magnetic force. The allowed values of ${L}_{z}$ are
where ${L}_{z}$ is the $z$-component of the angular momentum and ${m}_{l}$ is the angular momentum projection quantum number. The rule in parentheses for the values of ${m}_{l}$ is that it can range from $-l$ to $l$ in steps of one. For example, if $l=2$ , then ${m}_{l}$ can have the five values –2, –1, 0, 1, and 2. Each ${m}_{l}$ corresponds to a different energy in the presence of a magnetic field, so that they are related to the splitting of spectral lines into discrete parts, as discussed in the preceding section. If the $z$ -component of angular momentum can have only certain values, then the angular momentum can have only certain directions, as illustrated in [Figure 1].

Calculate the angles that the angular momentum vector $\vb{L}$ can make with the $z$ -axis for $l=1$ , as illustrated in [Figure 1].
Strategy
[Figure 1] represents the vectors $\vb{L}$ and ${\vb{L}}_{z}$ as usual, with arrows proportional to their magnitudes and pointing in the correct directions. $\vb{L}$ and ${\vb{L}}_{z}$ form a right triangle, with $\vb{L}$ being the hypotenuse and ${\vb{L}}_{z}$ the adjacent side. This means that the ratio of ${\vb{L}}_{z}$ to $\vb{L}$ is the cosine of the angle of interest. We can find $\vb{L}$ and ${\vb{L}}_{z}$ using $L=\sqrt{l\left(l+1\right)}\frac{h}{2\pi }$ and ${L}_{z}=m\frac{h}{2\pi }$.
Solution
We are given $l=1$ , so that ${m}_{l}$ can be +1, 0, or −1. Thus $L$ has the value given by $L=\sqrt{l\left(l+1\right)}\frac{h}{2\pi }$.
$L_{z}$ can have three values, given by $L_{z}= m_{l}\frac{h}{2\pi }$.
As can be seen in [Figure 1], $\cos \theta =\frac{ L_{z} }{L} ,$ and so for $m_{l}=+1$ , we have
Thus,
Similarly, for ${m}_{l}=0$ , we find $\cos {\theta }_{2}=0$ ; thus,
And for ${m}_{l}=-1$ ,
so that
Discussion
The angles are consistent with the figure. Only the angle relative to the $z$ -axis is quantized. $L$ can point in any direction as long as it makes the proper angle with the $z$ -axis. Thus the angular momentum vectors lie on cones as illustrated. This behavior is not observed on the large scale. To see how the correspondence principle holds here, consider that the smallest angle ( ${\theta }_{1}$ in the example) is for the maximum value of ${m}_{l}=0$ , namely ${m}_{l}=l$ . For that smallest angle,
which approaches 1 as $l$ becomes very large. If $\cos \theta =1$ , then $\theta =0º$ . Furthermore, for large $l$ , there are many values of ${m}_{l}$ , so that all angles become possible as $l$ gets very large.
There are two more quantum numbers of immediate concern. Both were first discovered for electrons in conjunction with fine structure in atomic spectra. It is now well established that electrons and other fundamental particles have _ intrinsic spin_, roughly analogous to a planet spinning on its axis. This spin is a fundamental characteristic of particles, and only one magnitude of intrinsic spin is allowed for a given type of particle. Intrinsic angular momentum is quantized independently of orbital angular momentum. Additionally, the direction of the spin is also quantized. It has been found that the magnitude of the intrinsic (internal) spin angular momentum, $S$ , of an electron is given by
where $s$ is defined to be the spin quantum number. This is very similar to the quantization of $L$ given in $L=\sqrt{l\left(l+1\right)}\frac{h}{2\pi }$ , except that the only value allowed for $s$ for electrons is 1/2.
The direction of intrinsic spin is quantized, just as is the direction of orbital angular momentum. The direction of spin angular momentum along one direction in space, again called the $z$ -axis, can have only the values
for electrons. ${S}_{z}$ is the ** $z$ -component of spin angular momentum** and ${m}_{s}$ is the spin projection quantum number. For electrons, $s$ can only be 1/2, and ${m}_{s}$ can be either +1/2 or -1/2. Spin projection ${m}_{s}=+1/2$ is referred to as spin up, whereas ${m}_{s}=-1/2$ is called spin down. These are illustrated in [Figure 5].
In later chapters, we will see that intrinsic spin is a characteristic of all subatomic particles. For some particles $s$ is half-integral, whereas for others $s$ is integral—there are crucial differences between half-integral spin particles and integral spin particles. Protons and neutrons, like electrons, have $s=1/2$ , whereas photons have $s=1$ , and other particles called pions have $s=0$ , and so on.
To summarize, the state of a system, such as the precise nature of an electron in an atom, is determined by its particular quantum numbers. These are expressed in the form $\left(\mathrm{n, l,} {m}_{l}, {m}_{s}\right)$ —see [Table 1] For electrons in atoms, the principal quantum number can have the values $n=1, 2, 3, ...$ . Once $n$ is known, the values of the angular momentum quantum number are limited to $l=1, 2, 3, ...,n-1$ . For a given value of $l$ , the angular momentum projection quantum number can have only the values ${m}_{l}=-l, -l +1, ...,-1, 0, 1, ..., l-1, l$ . Electron spin is independent of $\mathrm{n, l,}$ and ${m}_{l}$ , always having $s=1/2$ . The spin projection quantum number can have two values, ${m}\_{s}=1/2 \text{ or } -1/2$.
| Name | Symbol | Allowed values |
|---|---|---|
| Principal quantum number | $$n $$ | $$1, 2, 3, ... $$ |
| Angular momentum | $$l $$ | $$0, 1, 2, ...n-1 $$ |
| Angular momentum projection | $${m}_{l} $$ | $$-l, -l +1, ..., -1, 0, 1, ..., l-1, l \left(\text{ or } 0, ±1, ±2, ..., ±l\right) $$ |
| Spin1 | $$s $$ | $$1 \text{/2}\left(\text{electrons}\right) $$ |
| Spin projection | $${m}_{s} $$ | $$-1/2, +1/2 $$ |
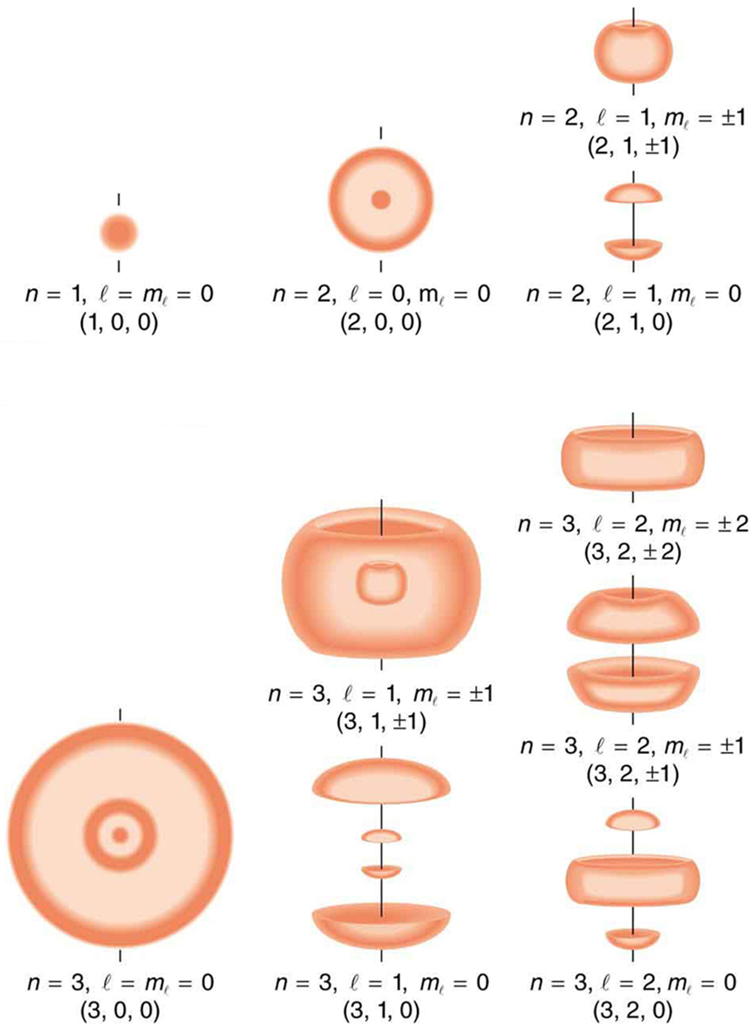
[Figure 2] shows several hydrogen states corresponding to different sets of quantum numbers. Note that these clouds of probability are the locations of electrons as determined by making repeated measurements—each measurement finds the electron in a definite location, with a greater chance of finding the electron in some places rather than others. With repeated measurements, the pattern of probability shown in the figure emerges. The clouds of probability do not look like nor do they correspond to classical orbits. The uncertainty principle actually prevents us and nature from knowing how the electron gets from one place to another, and so an orbit really does not exist as such. Nature on a small scale is again much different from that on the large scale.
We will see that the quantum numbers discussed in this section are valid for a broad range of particles and other systems, such as nuclei. Some quantum numbers, such as intrinsic spin, are related to fundamental classifications of subatomic particles, and they obey laws that will give us further insight into the substructure of matter and its interactions.
The classic Stern-Gerlach Experiment shows that atoms have a property called spin. Spin is a kind of intrinsic angular momentum, which has no classical counterpart. When the z-component of the spin is measured, one always gets one of two values: spin up or spin down.
Quantum numbers are used to express the allowed values of quantized entities. The principal quantum number $n$ labels the basic states of a system and is given by
The magnitude of angular momentum is given by
where $l$ is the angular momentum quantum number. The direction of angular momentum is quantized, in that its component along an axis defined by a magnetic field, called the $z$ -axis is given by
where ${L}_{z}$ is the $z$ -component of the angular momentum and ${m}_{l}$ is the angular momentum projection quantum number. Similarly, the electron’s intrinsic spin angular momentum $S$ is given by
$s$ is defined to be the spin quantum number. Finally, the direction of the electron’s spin along the $z$ -axis is given by
where ${S}_{z}$ is the $z$ -component of spin angular momentum and ${m}_{s}$ is the spin projection quantum number. Spin projection ${m}_{s}=+1/2$ is referred to as spin up, whereas ${m}_{s}=-1/2$ is called spin down. [Table 1] summarizes the atomic quantum numbers and their allowed values.
Define the quantum numbers $\mathrm{n, l,} {m}_{l}\mathrm{, s}$ , and ${m}_{s}$.
For a given value of $n$ , what are the allowed values of $l$ ?
For a given value of $l$ , what are the allowed values of ${m}_{l}$ ? What are the allowed values of ${m}_{l}$ for a given value of $n$ ? Give an example in each case.
List all the possible values of $s$ and ${m}_{s}$ for an electron. Are there particles for which these values are different? The same?
If an atom has an electron in the $n=5$ state with ${m}_{l}=3$ , what are the possible values of $l$ ?
Strategy
| We need to apply the rules for quantum numbers. The angular momentum quantum number $l$ must satisfy two constraints: (1) For a given principal quantum number $n$ , the allowed values of $l$ are $l = 0, 1, 2, ..., n-1$ ; and (2) The angular momentum projection quantum number $m_l$ is constrained by $$ | m_l | \le l $. We need to find which values of$ l $satisfy both conditions when$ n = 5 $and$ m_l = 3 $$ . |
Solution
Given that $n = 5$ , the first constraint tells us that:
Given that $m_l = 3$ , the second constraint requires:
Combining both constraints, we need $l \ge 3$ and $l \le 4$ (since $l < n = 5$ ).
Therefore, the possible values are: l = 3 or l = 4
If $l = 3$ (f-orbital), then $m_l$ can be –3, –2, –1, 0, +1, +2, or +3, so $m_l = 3$ is allowed.
If $l = 4$ (g-orbital), then $m_l$ can be –4, –3, –2, –1, 0, +1, +2, +3, or +4, so $m_l = 3$ is also allowed.
Discussion
This problem demonstrates the hierarchical relationship between quantum numbers in atoms. The principal quantum number $n = 5$ places an upper limit on $l$ ( $l \le 4$ ), while the value of $m_l = 3$ places a lower limit on $l$ ( $l \ge 3$ ). The fact that $m_l = 3$ rules out $l = 0, 1, 2$ is physically reasonable: $m_l$ represents the component of angular momentum along a specific axis, and this component cannot exceed the total angular momentum magnitude (which depends on $l$ ). In spectroscopic notation, $l = 3$ corresponds to an f-orbital (named historically: s, p, d, f for $l = 0, 1, 2, 3$ ), and $l = 4$ corresponds to a g-orbital. These higher angular momentum states first appear in atoms with many electrons. For example, f-orbitals begin to fill in the lanthanide elements (starting with cerium, element 58), while g-orbitals are not occupied in the ground states of known elements but can be accessed in excited states. Understanding which quantum number combinations are allowed is crucial for predicting electron configurations, chemical bonding, and spectroscopic properties of atoms.
An atom has an electron with ${m}_{l}=2$ . What is the smallest value of $n$ for this electron?
Strategy
| The angular momentum projection quantum number $m_l$ is constrained by the angular momentum quantum number $l$ through the relationship $$ | m_l | \le l $. Furthermore, for a given principal quantum number$ n $, the angular momentum quantum number can only take values$ l = 0, 1, 2, …, n-1 $. Therefore, we need to find the minimum value of$ n $such that$ l $can be large enough to accommodate$ m_l = 2 $$ . |
Solution
| Since $m_l = 2$ , we need $l \ge 2$ (because $$ | m_l | \le l $$ ). |
The minimum value of $l$ that satisfies this is $l = 2$ .
For $l = 2$ to be an allowed value, we need $l \le n - 1$ , which gives us $2 \le n - 1$ , or $n \ge 3$ .
Therefore, the smallest value of $n$ is n = 3.
Discussion
This problem illustrates the hierarchical relationship among the quantum numbers in atoms. The principal quantum number $n$ determines the energy level and sets an upper limit on the angular momentum quantum number $l$ . In turn, $l$ determines the magnitude of the orbital angular momentum and sets limits on the possible values of $m_l$ , which specifies the orientation of the angular momentum vector. For $n = 3$ , the allowed values of $l$ are 0, 1, and 2. When $l = 2$ (called a d-orbital), $m_l$ can take any of the five values: –2, –1, 0, +1, or +2. Each of these corresponds to a different orientation of the electron’s orbital angular momentum relative to an external magnetic field. The fact that we need at least $n = 3$ for $m_l = 2$ explains why d-orbitals first appear in the third shell of atoms (the M shell), which is crucial for understanding the electron configurations of transition metals and their chemical properties.
What are the possible values of ${m}_{l}$ for an electron in the $n=4$ state?
Strategy
For a given principal quantum number $n$ , the angular momentum quantum number $l$ can range from 0 to $n-1$ . For each value of $l$ , the angular momentum projection quantum number $m_l$ can range from $-l$ to $+l$ in integer steps. We need to determine all possible values of $m_l$ for any allowed $l$ when $n = 4$ .
Solution
For $n = 4$ , the allowed values of $l$ are:
For each value of $l$ , the allowed values of $m_l$ are:
For l = 0 (4s orbital):
For l = 1 (4p orbitals):
For l = 2 (4d orbitals):
For l = 3 (4f orbitals):
Combining all possibilities, the complete set of possible values of $m_l$ for $n = 4$ is:
Or more compactly: $m_l = 0, \pm 1, \pm 2, \pm 3$
Discussion
| This problem reveals an important feature of atomic structure: for a given principal quantum number $n$ , there are multiple orbitals with different angular momentum properties. The $n = 4$ shell contains 4s (1 orbital), 4p (3 orbitals), 4d (5 orbitals), and 4f (7 orbitals), for a total of 16 orbitals. Since each orbital can hold two electrons (with opposite spins), the $n = 4$ shell can accommodate up to 32 electrons. The seven distinct values of $m_l$ ( –3 to +3 ) represent different orientations of the orbital angular momentum vector relative to an external magnetic field (the $z$ -axis). In the absence of an external field, orbitals with the same $n$ and $l$ but different $m_l$ values have the same energy (they are “degenerate”). However, when an external magnetic field is applied, this degeneracy is broken—the Zeeman effect causes each $m_l$ value to correspond to a slightly different energy. This is why spectral lines split in magnetic fields. The maximum value of $$ | m_l | $equals the maximum value of$ l $for that shell, which is$ n - 1 $$ . Understanding these relationships is essential for explaining atomic spectra, magnetic properties of atoms, and the structure of the periodic table. |
What, if any, constraints does a value of ${m}_{l}=1$ place on the other quantum numbers for an electron in an atom?
Strategy
| We need to examine the relationships between the quantum numbers to determine what constraints $m_l = 1$ places on the other quantum numbers ( $n$ , $l$ , and $m_s$ ). The key relationships are: (1) $$ | m_l | \le l $, (2)$ l \le n - 1 $, and (3)$ m_s $$ is independent of the orbital quantum numbers. |
Solution
Given that $m_l = 1$ :
| Constraint on l: Since $$ | m_l | \le l $, we must have$ l \ge 1 $. Therefore, the electron cannot be in an s-orbital ($ l = 0 $) but could be in a p-orbital ($ l = 1 $), d-orbital ($ l = 2 $), f-orbital ($ l = 3 $$ ), or any higher angular momentum state. |
Constraint on n: Since $l \ge 1$ and $l \le n - 1$ , we need $n - 1 \ge 1$ , which gives $n \ge 2$ . Therefore, the electron must be in the second shell or higher ( $n = 2, 3, 4, ...$ ).
Constraint on $m_s$: The spin projection quantum number $m_s$ can be either +1/2 or –1/2, independent of the value of $m_l$ . There is no constraint on $m_s$ .
In summary:
Discussion
This problem demonstrates the interconnected nature of quantum numbers in atoms. The value of $m_l = 1$ tells us about the orientation of the electron’s orbital angular momentum but doesn’t uniquely specify the state of the electron. For example, the electron could be in a 2p orbital ( $n = 2, l = 1$ ), a 3p orbital ( $n = 3, l = 1$ ), a 3d orbital ( $n = 3, l = 2$ ), or many other possibilities. Each of these states would have different energies and different probability distributions for the electron’s position. The fact that $m_l = 1$ excludes only s-orbitals ( $l = 0$ ) but allows all others shows that $m_l$ specifies orientation rather than the magnitude of angular momentum. The independence of $m_s$ reflects the fact that electron spin is an intrinsic property not related to orbital motion. Understanding these constraints is essential for writing electron configurations and predicting the chemical and spectroscopic properties of atoms.
(a) Calculate the magnitude of the angular momentum for an $l=1$ electron. (b) Compare your answer to the value Bohr proposed for the $n=1$ state.
Strategy
(a) The magnitude of angular momentum is given by the quantum mechanical formula $L = \sqrt{l(l+1)} \frac{h}{2\pi}$ , where $l$ is the angular momentum quantum number and $h = 6.626 \times 10^{-34} \text{ J·s}$ is Planck’s constant. We’ll substitute $l = 1$ and calculate.
(b) Bohr’s original model proposed that the angular momentum of an electron in orbit is quantized as $L = n\frac{h}{2\pi}$ , where $n$ is the principal quantum number. For the $n = 1$ state (ground state), Bohr predicted $L = \frac{h}{2\pi}$ . We’ll calculate this value and compare it to the quantum mechanical result.
Solution
(a) For $l = 1$ :
(b) Bohr’s value for the $n = 1$ state:
Comparing the two values:
The quantum mechanical value for $l = 1$ is $\sqrt{2}$ times larger than Bohr’s prediction for $n = 1$ .
Discussion
This comparison reveals a fundamental difference between Bohr’s semi-classical model and the full quantum mechanical treatment of the atom. Bohr correctly predicted that angular momentum is quantized, but his specific formula $L = n\frac{h}{2\pi}$ turned out to be incorrect. Modern quantum mechanics shows that (1) the magnitude of angular momentum depends on $l$ , not $n$ , and follows $L = \sqrt{l(l+1)} \frac{h}{2\pi}$ ; and (2) for a given $n$ , $l$ can range from 0 to $n-1$ , meaning different orbitals in the same shell have different angular momenta.
Most surprisingly, quantum mechanics reveals that the ground state of hydrogen ( $n = 1, l = 0$ ) has zero angular momentum, not $\frac{h}{2\pi}$ as Bohr proposed! This is completely incompatible with Bohr’s picture of electrons orbiting in circular paths. Instead, the $l = 0$ state (s-orbital) is spherically symmetric with no orbital angular momentum at all—the electron’s probability cloud is centered on the nucleus with no preferred direction.
The $l = 1$ state we calculated represents a p-orbital, which has angular momentum and a more complex spatial structure. The factor $\sqrt{l(l+1)}$ rather than just $l$ appears because of the way angular momentum operators work in quantum mechanics—this same factor appears for all quantized angular momenta. The comparison shows that while Bohr’s model was revolutionary and correct in predicting quantization, the details of quantum mechanics are more subtle and accurate. Nevertheless, Bohr’s achievement in introducing quantization to atomic physics was a crucial step toward the modern theory.
(a) What is the magnitude of the angular momentum for an $l=1$ electron? (b) Calculate the magnitude of the electron’s spin angular momentum. (c) What is the ratio of these angular momenta?
Strategy
We’ll use the formulas for quantized angular momentum. For orbital angular momentum: $L = \sqrt{l(l+1)} \frac{h}{2\pi}$ . For spin angular momentum: $S = \sqrt{s(s+1)} \frac{h}{2\pi}$ , where $s = 1/2$ for electrons. We’ll calculate both values and then find their ratio.
Solution
(a) For $l = 1$ , the magnitude of the orbital angular momentum is:
Substituting $h = 6.626 \times 10^{-34} \text{ J·s}$ :
(b) For electron spin with $s = 1/2$ :
(c) The ratio of orbital to spin angular momentum is:
Discussion
This calculation reveals several important aspects of atomic structure. First, both orbital and spin angular momenta are on the order of $10^{-34} \text{ J·s}$ , which is the same order as Planck’s constant divided by $2\pi$ (often written as $\hbar$ ). This demonstrates that atomic-scale angular momentum is quantized in units of $\hbar$ . Second, the ratio of about 1.63 shows that for an $l = 1$ electron (a p-orbital), the orbital angular momentum is somewhat larger than the spin angular momentum, but they are of the same order of magnitude. This comparability is important because both contribute to the total angular momentum of the atom and both interact with external magnetic fields. The spin-orbit coupling—the interaction between the electron’s orbital and spin magnetic moments—leads to fine structure in atomic spectra. For different values of $l$ , this ratio changes: for $l = 3$ (next problem), the ratio is 4, showing that as $l$ increases, orbital angular momentum dominates over spin. Understanding these relationships is crucial for explaining magnetic properties of atoms, spectroscopic splitting patterns, and the behavior of atoms in external magnetic fields.
Repeat [the previous exercise] for $l=3$.
Strategy
Following the same approach as Exercise 7, we’ll calculate: (a) the magnitude of orbital angular momentum for $l = 3$ using $L = \sqrt{l(l+1)} \frac{h}{2\pi}$ ; (b) the magnitude of electron spin angular momentum using $S = \sqrt{s(s+1)} \frac{h}{2\pi}$ where $s = 1/2$ ; (c) the ratio of these two quantities.
Solution
(a) For $l = 3$ , the magnitude of orbital angular momentum is:
(b) The electron spin angular momentum (same for all electrons regardless of $l$ ):
(c) The ratio of orbital to spin angular momentum:
The ratio is 4.
Discussion
Comparing this result to Exercise 7 (where $l = 1$ gave a ratio of 1.63), we see that as $l$ increases, the orbital angular momentum becomes much larger relative to the spin angular momentum. For $l = 3$ (an f-orbital), the orbital angular momentum is exactly 4 times the spin angular momentum, whereas for $l = 1$ (a p-orbital) it was only 1.63 times larger. This makes sense because orbital angular momentum scales as $\sqrt{l(l+1)}$ , which grows roughly as $\sqrt{l^2} = l$ for large $l$ , while spin angular momentum is fixed at $\sqrt{3/4} \frac{h}{2\pi}$ for all electrons. The $l = 3$ state corresponds to f-orbitals, which first appear in the fourth shell (n = 4) and are important in the chemistry of lanthanide and actinide elements. The large orbital angular momentum of f-electrons contributes significantly to the magnetic properties of these elements. Both orbital and spin angular momenta couple together to give the total angular momentum of the atom, and this coupling is responsible for fine structure in atomic spectra and for the magnetic moments of atoms. The fact that orbital angular momentum can vary widely (depending on $l$ ) while spin is always the same ($s = 1/2$ for electrons) means that the relative importance of spin-orbit coupling varies greatly for different orbitals—it’s more important for s and p electrons (low $l$ ) than for d and f electrons (high $l$ ) in terms of the fractional contribution.
(a) How many angles can $L$ make with the $z$-axis for an $l=2$ electron? (b) Calculate the value of the smallest angle.
Strategy
For a given value of $l$ , the angular momentum projection quantum number $m_l$ can take values from $-l$ to $+l$ in integer steps, giving $2l + 1$ possible values. Each value of $m_l$ corresponds to a different angle that the angular momentum vector $\mathbf{L}$ makes with the $z$ -axis. The angle can be found from $\cos \theta = \frac{L_z}{L} = \frac{m_l}{\sqrt{l(l+1)}}$ .
Solution
(a) For $l = 2$ , the possible values of $m_l$ are:
This gives $2l + 1 = 2(2) + 1 = 5$ possible orientations. Therefore, $\mathbf{L}$ can make 5 different angles with the $z$ -axis.
(b) The smallest angle occurs when $m_l$ has its maximum value, $m_l = +2$ (or minimum value $m_l = -2$ , which gives the same angle by symmetry). The magnitude of $\mathbf{L}$ is:
The $z$ -component is:
The angle is given by:
The smallest angle is 35.3° (or 0.616 radians).
Discussion
This problem illustrates the concept of space quantization—the remarkable fact that the angular momentum vector can only point in certain discrete directions relative to an external reference direction (the $z$ -axis, typically defined by an external magnetic field). For $l = 2$ (a d-orbital), the five allowed angles correspond to the five d-orbitals in chemistry ( $d_{z^2}, d_{xz}, d_{yz}, d_{xy}, d_{x^2-y^2}$ ). The smallest angle of 35.3° represents the most closely aligned orientation—even at this “closest” alignment, the angular momentum vector cannot point directly along the $z$ -axis. This is fundamentally different from classical physics, where a rotating object could have its angular momentum pointing in any direction. The fact that even the maximum value of $m_l = l$ doesn’t allow perfect alignment ( $\theta = 0°$ ) is due to the quantum mechanical relationship $L = \sqrt{l(l+1)} \frac{h}{2\pi}$ rather than $l\frac{h}{2\pi}$ . This “ $\sqrt{l(l+1)}$ “ factor, rather than just “ $l$ ,” ensures that $L > L_z$ for all states, preventing perfect alignment and reflecting the uncertainty principle’s constraint on simultaneously knowing all components of angular momentum.
What angles can the spin $S$ of an electron make with the $z$ -axis?
Strategy
For electron spin, the spin quantum number is $s = 1/2$ , and the spin projection quantum number $m_s$ can be either $+1/2$ (spin up) or $-1/2$ (spin down). The magnitude of the spin angular momentum is $S = \sqrt{s(s+1)} \frac{h}{2\pi}$ , and its $z$ -component is $S_z = m_s \frac{h}{2\pi}$ . The angle between $\mathbf{S}$ and the $z$ -axis can be found from $\cos \theta = \frac{S_z}{S}$ .
Solution
The magnitude of electron spin is:
The $z$ -component can have two values:
For $m_s = +1/2$ (spin up):
For $m_s = -1/2$ (spin down):
The electron spin can make only two angles with the $z$ -axis: 54.7° and 125.3°.
Discussion
This result demonstrates the quantization of spin direction—a purely quantum mechanical phenomenon with no classical analogue. Unlike orbital angular momentum, where the number of allowed angles depends on $l$ (giving $2l+1$ orientations), electron spin always has exactly two allowed orientations regardless of the atom or state. These two orientations are commonly called “spin up” and “spin down,” though these names are somewhat misleading since the spin is never aligned parallel or antiparallel to the $z$ -axis.
| Notice that even for the “spin up” state ( $m_s = +1/2$ ), the angle is 54.7° rather than 0°—the spin cannot point directly along the $z$ -axis. This is a consequence of the $\sqrt{s(s+1)}$ factor in the magnitude formula: since $S = \frac{\sqrt{3}}{2} \frac{h}{2\pi}$ is always larger than $$ | S_z | = \frac{1}{2} \frac{h}{2\pi} $$ , perfect alignment is impossible. This reflects a fundamental aspect of quantum mechanics: you cannot simultaneously know all three components of angular momentum (Heisenberg uncertainty principle for angular momentum). |
The two angles are symmetric about 90°: one is 90° – 35.3° = 54.7°, and the other is 90° + 35.3° = 125.3°. This symmetry reflects the fact that the two spin states have equal magnitude angular momenta, just pointing in generally opposite directions.
Electron spin is fundamental to understanding: (1) the Pauli exclusion principle—two electrons in the same orbital must have opposite spins; (2) magnetic properties of materials—unpaired electron spins create magnetic moments; (3) fine structure in atomic spectra—spin-orbit coupling splits spectral lines; (4) modern technologies like MRI, which depends on nuclear spin (similar to electron spin but for protons), and spintronics, which exploits electron spin for information storage and processing.