
The great Danish physicist Niels Bohr (1885–1962) made immediate use of Rutherford’s planetary model of the atom. ([Figure 1]). Bohr became convinced of its validity and spent part of 1912 at Rutherford’s laboratory. In 1913, after returning to Copenhagen, he began publishing his theory of the simplest atom, hydrogen, based on the planetary model of the atom. For decades, many questions had been asked about atomic characteristics. From their sizes to their spectra, much was known about atoms, but little had been explained in terms of the laws of physics. Bohr’s theory explained the atomic spectrum of hydrogen and established new and broadly applicable principles in quantum mechanics.

As noted in Quantization of Energy , the energies of some small systems are quantized. Atomic and molecular emission and absorption spectra have been known for over a century to be discrete (or quantized). ( See [Figure 2].) Maxwell and others had realized that there must be a connection between the spectrum of an atom and its structure, something like the resonant frequencies of musical instruments. But, in spite of years of efforts by many great minds, no one had a workable theory. (It was a running joke that any theory of atomic and molecular spectra could be destroyed by throwing a book of data at it, so complex were the spectra.) Following Einstein’s proposal of photons with quantized energies directly proportional to their wavelengths, it became even more evident that electrons in atoms can exist only in discrete orbits.
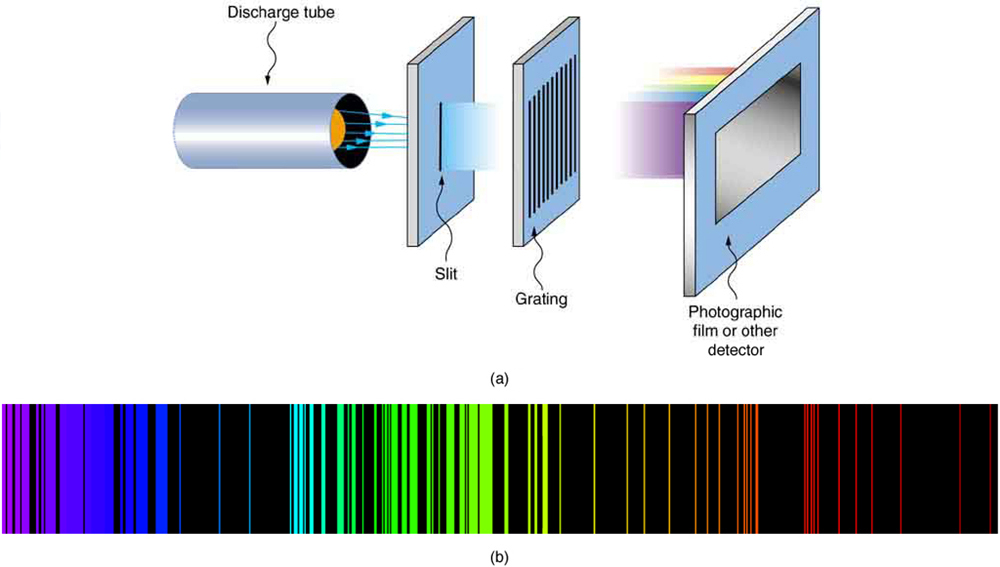
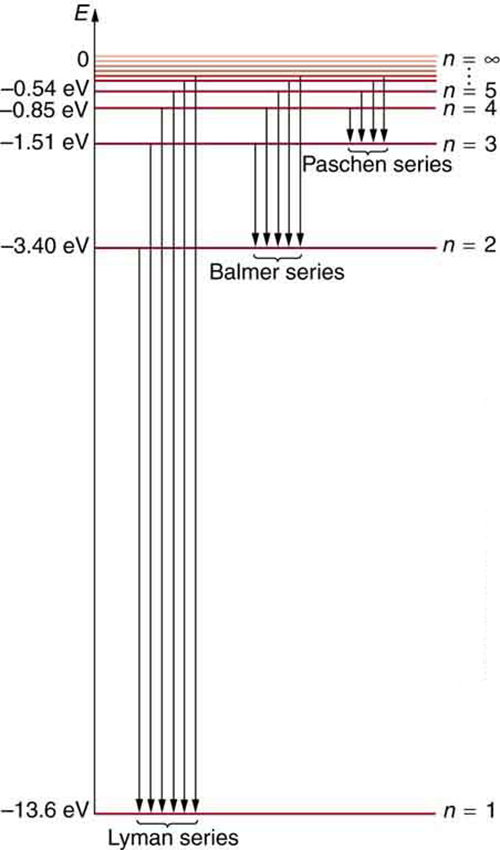
In some cases, it had been possible to devise formulas that described the emission spectra. As you might expect, the simplest atom—hydrogen, with its single electron—has a relatively simple spectrum. The hydrogen spectrum had been observed in the infrared (IR), visible, and ultraviolet (UV), and several series of spectral lines had been observed. (See [Figure 3].) These series are named after early researchers who studied them in particular depth.
The observed hydrogen-spectrum wavelengths can be calculated using the following formula:
where $\lambda$ is the wavelength of the emitted EM radiation and $R$ is the Rydberg constant, determined by the experiment to be
The constant ${n}_{\text{f}}$ is a positive integer associated with a specific series. For the Lyman series, ${n}_{\text{f}}=1$ ; for the Balmer series, ${n}_{\text{f}}=2$ ; for the Paschen series, ${n}_{\text{f}}=3$ ; and so on. The Lyman series is entirely in the UV, while part of the Balmer series is visible with the remainder UV. The Paschen series and all the rest are entirely IR. There are apparently an unlimited number of series, although they lie progressively farther into the infrared and become difficult to observe as ${n}_{\text{f}}$ increases. The constant ${n}_{\text{i}}$ is a positive integer, but it must be greater than ${n}_{\text{f}}$ . Thus, for the Balmer series, ${n}_{\text{f}}=2$ and ${n}_{\text{i}}=3, 4, 5, 6, ...$ . Note that ${n}_{\text{i}}$ can approach infinity. While the formula in the wavelengths equation was just a recipe designed to fit data and was not based on physical principles, it did imply a deeper meaning. Balmer first devised the formula for his series alone, and it was later found to describe all the other series by using different values of ${n}_{\text{f}}$ . Bohr was the first to comprehend the deeper meaning. Again, we see the interplay between experiment and theory in physics. Experimentally, the spectra were well established, an equation was found to fit the experimental data, but the theoretical foundation was missing.
What is the distance between the slits of a grating that produces a first-order maximum for the second Balmer line at an angle of $15 ^\circ$ ?
Strategy and Concept
For an Integrated Concept problem, we must first identify the physical principles involved. In this example, we need to know (a) the wavelength of light as well as (b) conditions for an interference maximum for the pattern from a double slit. Part (a) deals with a topic of the present chapter, while part ( b) considers the wave interference material of Wave Optics.
Solution for (a)
Hydrogen spectrum wavelength. The Balmer series requires that ${n}_{\text{f}}=2$ . The first line in the series is taken to be for ${n}_{\text{i}}=3$ , and so the second would have ${n}_{\text{i}}=4$. The calculation is a straightforward application of the wavelength equation. Entering the determined values for ${n}_{\text{f}}$ and ${n}_{\text{i}}$ yields
Inverting to find $\lambda$ gives
Discussion for (a)
This is indeed the experimentally observed wavelength, corresponding to the second (blue-green) line in the Balmer series. More impressive is the fact that the same simple recipe predicts all of the hydrogen spectrum lines, including new ones observed in subsequent experiments. What is nature telling us?
Solution for (b)
Double-slit interference (Wave Optics). To obtain constructive interference for a double slit, the path length difference from two slits must be an integral multiple of the wavelength. This condition was expressed by the equation
where $d$ is the distance between slits and $\theta$ is the angle from the original direction of the beam. The number $m$ is the order of the interference; $m=1$ in this example. Solving for $d$ and entering known values yields
Discussion for (b)
This number is similar to those used in the interference examples of Introduction to Quantum Physics (and is close to the spacing between slits in commonly used diffraction glasses).
Bohr was able to derive the formula for the hydrogen spectrum using basic physics, the planetary model of the atom, and some very important new proposals. His first proposal is that only certain orbits are allowed: we say that the orbits of electrons in atoms are quantized. Each orbit has a different energy, and electrons can move to a higher orbit by absorbing energy and drop to a lower orbit by emitting energy. If the orbits are quantized, the amount of energy absorbed or emitted is also quantized, producing discrete spectra. Photon absorption and emission are among the primary methods of transferring energy into and out of atoms. The energies of the photons are quantized, and their energy is explained as being equal to the change in energy of the electron when it moves from one orbit to another. In equation form, this is
Here, $\Delta E$ is the change in energy between the initial and final orbits, and $hf$ is the energy of the absorbed or emitted photon. It is quite logical (that is, expected from our everyday experience) that energy is involved in changing orbits. A blast of energy is required for the space shuttle, for example, to climb to a higher orbit. What is not expected is that atomic orbits should be quantized. This is not observed for satellites or planets, which can have any orbit given the proper energy. ( See [Figure 4].)
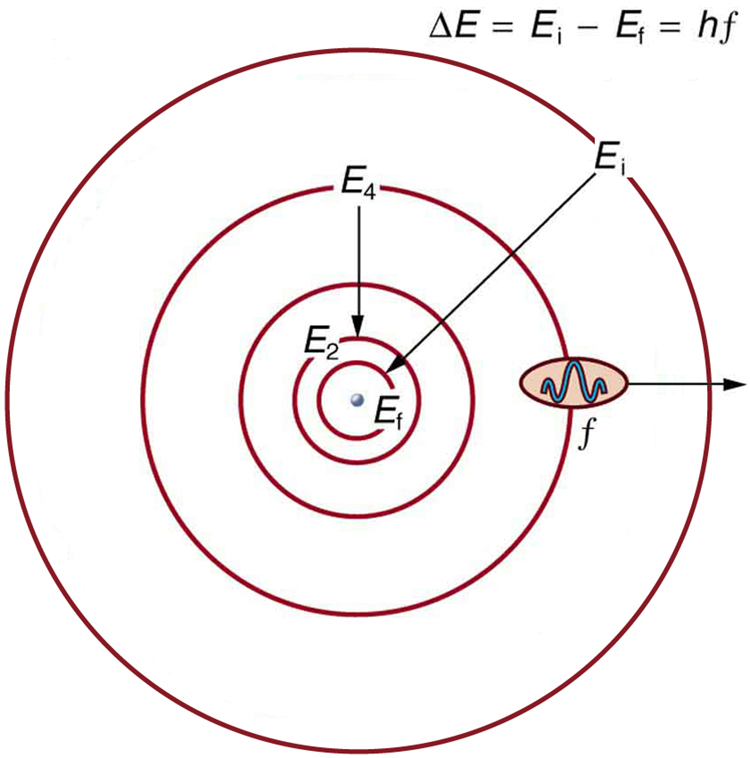
[Figure 5] shows an energy-level diagram, a convenient way to display energy states. In the present discussion, we take these to be the allowed energy levels of the electron. Energy is plotted vertically with the lowest or ground state at the bottom and with excited states above. Given the energies of the lines in an atomic spectrum, it is possible (although sometimes very difficult) to determine the energy levels of an atom. Energy-level diagrams are used for many systems, including molecules and nuclei. A theory of the atom or any other system must predict its energies based on the physics of the system.
Bohr was clever enough to find a way to calculate the electron orbital energies in hydrogen. This was an important first step that has been improved upon, but it is well worth repeating here, because it does correctly describe many characteristics of hydrogen. Assuming circular orbits, Bohr proposed that the angular momentum $L$ of an electron in its orbit is quantized, that is, it has only specific, discrete values. The value for $L$ is given by the formula
where $L$ is the angular momentum, ${m}_{e}$ is the electron’s mass, ${r}\_{n}$ is the radius of the $n$ th orbit, and $h$ is Planck’s constant. Note that angular momentum is $L=\mathrm{I\omega }$ . For a small object at a radius $r, I={\text{mr}}^{2}$ and $\omega =v/r$ , so that $L=\left({\text{mr}}^{2}\right)\left(v/r\right)=\text{mvr}$ . Quantization says that this value of $\text{mvr}$ can only be equal to $h/2, 2h/2, 3h/2$ , etc. At the time, Bohr himself did not know why angular momentum should be quantized, but using this assumption he was able to calculate the energies in the hydrogen spectrum, something no one else had done at the time.
From Bohr’s assumptions, we will now derive a number of important properties of the hydrogen atom from the classical physics we have covered in the text. We start by noting the centripetal force causing the electron to follow a circular path is supplied by the Coulomb force. To be more general, we note that this analysis is valid for any single-electron atom. So, if a nucleus has $Z$ protons ( $Z=1$ for hydrogen, 2 for helium, etc.) and only one electron, that atom is called a hydrogen-like atom. The spectra of hydrogen-like ions are similar to hydrogen, but shifted to higher energy by the greater attractive force between the electron and nucleus. The magnitude of the centripetal force is ${m}_{e}{v}^{2}/{r}_{n}$ , while the Coulomb force is $k\left( {\text{Zq}}_{e}\right)\left({q}_{e}\right)/{r}_{n}^{2}$ . The tacit assumption here is that the nucleus is more massive than the stationary electron, and the electron orbits about it. This is consistent with the planetary model of the atom. Equating these,
Angular momentum quantization is stated in an earlier equation. We solve that equation for $v$ , substitute it into the above, and rearrange the expression to obtain the radius of the orbit. This yields:
where ${a}_{\text{B}}$ is defined to be the Bohr radius, since for the lowest orbit $\left(n=1\right)$ and for hydrogen $\left(Z=1\right)$ , ${r}_{1}={a}_{\text{B}}$ . It is left for this chapter’s Problems and Exercises to show that the Bohr radius is
These last two equations can be used to calculate the radii of the allowed ( quantized) electron orbits in any hydrogen-like atom. It is impressive that the formula gives the correct size of hydrogen, which is measured experimentally to be very close to the Bohr radius. The earlier equation also tells us that the orbital radius is proportional to ${n}^{2}$ , as illustrated in [Figure 6].
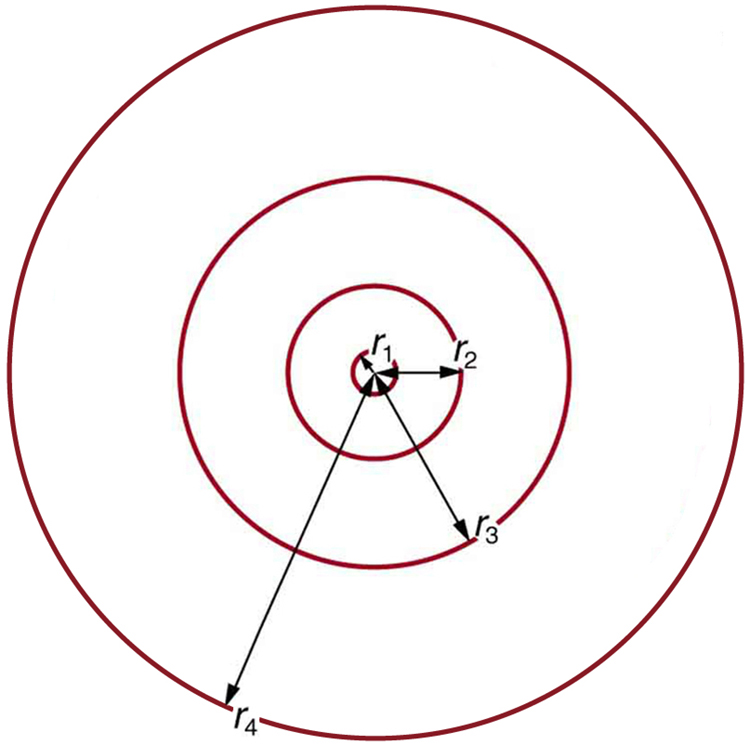
To get the electron orbital energies, we start by noting that the electron energy is the sum of its kinetic and potential energy:
Kinetic energy is the familiar $\text{KE}=\left(1/2\right){m}_{e}{v}^{2}$ , assuming the electron is not moving at relativistic speeds. Potential energy for the electron is electrical, or $\text{PE}={q}_{e}V$ , where $V$ is the potential due to the nucleus, which looks like a point charge. The nucleus has a positive charge $Zq_{e}$ ; thus, $V= kZq_{e}/{r}_{n}$ , recalling an earlier equation for the potential due to a point charge. Since the electron’s charge is negative, we see that $\text{PE}=-kZq_{e}/{r}_{n}$ . Entering the expressions for $\text{KE}$ and $\text{PE}$ , we find
Now we substitute ${r}_{n}$ and $v$ from earlier equations into the above expression for energy. Algebraic manipulation yields
for the orbital energies of hydrogen-like atoms. Here, ${E}_{0}$ is the ground-state energy $\left(n=1\right)$ for hydrogen $\left(Z=1\right)$ and is given by
Thus, for hydrogen,
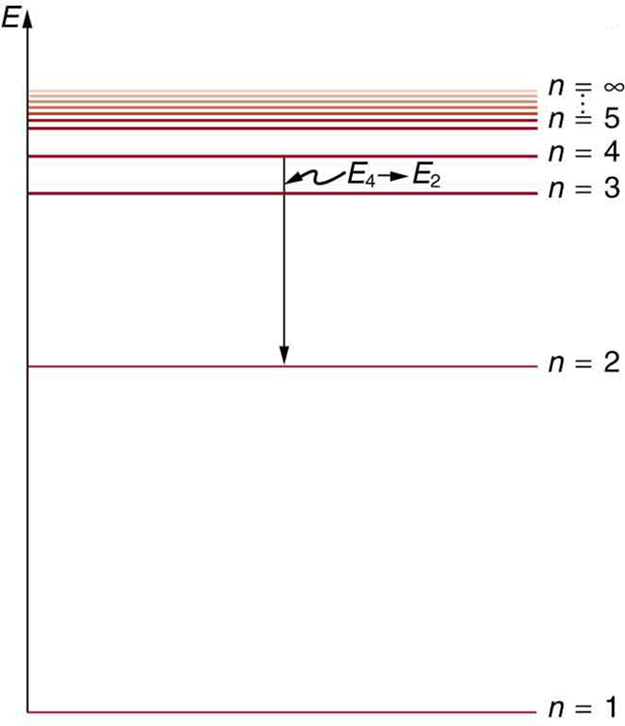
[Figure 7] shows an energy-level diagram for hydrogen that also illustrates how the various spectral series for hydrogen are related to transitions between energy levels.
Electron total energies are negative, since the electron is bound to the nucleus, analogous to being in a hole without enough kinetic energy to escape. As $n$ approaches infinity, the total energy becomes zero. This corresponds to a free electron with no kinetic energy, since ${r}_{n}$ gets very large for large $n$ , and the electric potential energy thus becomes zero. Thus, 13.6 eV is needed to ionize hydrogen (to go from –13.6 eV to 0, or unbound), an experimentally verified number. Given more energy, the electron becomes unbound with some kinetic energy. For example, giving 15.0 eV to an electron in the ground state of hydrogen strips it from the atom and leaves it with 1.4 eV of kinetic energy.
Finally, let us consider the energy of a photon emitted in a downward transition, given by the equation to be
Substituting ${E}_{n}=\left(-13.6 \text{eV}/{n}^{2}\right)$ , we see that
Dividing both sides of this equation by $hc$ gives an expression for $1/\lambda$ :
It can be shown that
is the Rydberg constant. Thus, we have used Bohr’s assumptions to derive the formula first proposed by Balmer years earlier as a recipe to fit experimental data.
We see that Bohr’s theory of the hydrogen atom answers the question as to why this previously known formula describes the hydrogen spectrum. It is because the energy levels are proportional to $1/{n}^{2}$ , where $n$ is a non-negative integer. A downward transition releases energy, and so ${n}_{i}$ must be greater than ${n}_{\text{f}}$ . The various series are those where the transitions end on a certain level. For the Lyman series, ${n}_{\text{f}}=1$ — that is, all the transitions end in the ground state (see also [Figure 7]). For the Balmer series, ${n}_{\text{f}}=2$ , or all the transitions end in the first excited state; and so on. What was once a recipe is now based in physics, and something new is emerging—angular momentum is quantized.
Bohr did what no one had been able to do before. Not only did he explain the spectrum of hydrogen, he correctly calculated the size of the atom from basic physics. Some of his ideas are broadly applicable. Electron orbital energies are quantized in all atoms and molecules. Angular momentum is quantized. The electrons do not spiral into the nucleus, as expected classically (accelerated charges radiate, so that the electron orbits classically would decay quickly, and the electrons would sit on the nucleus—matter would collapse). These are major triumphs.
But there are limits to Bohr’s theory. It cannot be applied to multielectron atoms, even one as simple as a two-electron helium atom. Bohr’s model is what we call semiclassical. The orbits are quantized (nonclassical) but are assumed to be simple circular paths (classical). As quantum mechanics was developed, it became clear that there are no well-defined orbits; rather, there are clouds of probability. Bohr’s theory also did not explain that some spectral lines are doublets (split into two) when examined closely. We shall examine many of these aspects of quantum mechanics in more detail, but it should be kept in mind that Bohr did not fail. Rather, he made very important steps along the path to greater knowledge and laid the foundation for all of atomic physics that has since evolved.
How did scientists figure out the structure of atoms without looking at them? Try out different models by shooting light at the atom. Check how the prediction of the model matches the experimental results.
where $\lambda$ is the wavelength of the emitted EM radiation and $R$ is the Rydberg constant, which has the value
The constants ${n}_{i}$ and ${n}_{f}$ are positive integers, and ${n}_{i}$ must be greater than ${n}_{f}$ .
where $\Delta E$ is the change in energy between the initial and final orbits and $hf$ is the energy of an absorbed or emitted photon. It is useful to plot orbital energies on a vertical graph called an energy-level diagram.
where $L$ is the angular momentum, ${r}_{n}$ is the radius of the $n\text{th}$ orbit, and $h$ is Planck’s constant. For all one-electron (hydrogen-like) atoms, the radius of an orbit is given by
$Z$ is the atomic number of an element (the number of electrons is has when neutral) and ${a}_{\text{B}}$ is defined to be the Bohr radius, which is
where ${E}_{0}$ is the ground-state energy and is given by
Thus, for hydrogen,
How do the allowed orbits for electrons in atoms differ from the allowed orbits for planets around the sun? Explain how the correspondence principle applies here.
Explain how Bohr’s rule for the quantization of electron orbital angular momentum differs from the actual rule.
What is a hydrogen-like atom, and how are the energies and radii of its electron orbits related to those in hydrogen?
By calculating its wavelength, show that the first line in the Lyman series is UV radiation.
Strategy
The Lyman series corresponds to transitions ending at the ground state, so $n_{\text{f}} = 1$ . The first line in the series corresponds to a transition from $n_i = 2$ to $n_f = 1$ . We’ll use the Rydberg formula to calculate the wavelength and determine if it falls in the UV region (wavelengths less than 400 nm).
Solution
Using the Rydberg formula:
With $n_{\text{f}} = 1$ and $n_i = 2$ :
Since 122 nm is much less than 400 nm (the lower limit of visible light), this is indeed UV radiation.
Discussion
The entire Lyman series lies in the ultraviolet region because these transitions all end at the ground state (n = 1), involving relatively large energy changes. The first line at 122 nm (also called Lyman-alpha) is the longest wavelength and lowest energy transition in this series. As we move to higher lines in the series (from n = 3, 4, 5, … to n = 1), the wavelengths get progressively shorter, approaching the series limit at 91.2 nm as n approaches infinity. This UV radiation from hydrogen is important in astronomy—Lyman-alpha is one of the most studied spectral lines in astrophysics, used to probe distant galaxies, intergalactic medium, and the early universe. However, because Earth’s atmosphere absorbs UV radiation, these lines must be observed from space-based telescopes.
Find the wavelength of the third line in the Lyman series, and identify the type of EM radiation.
Strategy
The Lyman series corresponds to transitions ending at the ground state, so $n_{\text{f}} = 1$ . The first line in the series corresponds to a transition from $n_i = 2$ to $n_f = 1$ , the second line from $n_i = 3$ to $n_f = 1$ , and the third line from $n_i = 4$ to $n_f = 1$ . We’ll use the Rydberg formula:
where $R = 1.097 \times 10^7 \text{ m}^{-1}$ is the Rydberg constant.
Solution
For the third line in the Lyman series, we have $n_{\text{f}} = 1$ and $n_i = 4$ :
This wavelength of 97.2 nm is in the ultraviolet (UV) region of the electromagnetic spectrum.
Discussion
The entire Lyman series lies in the UV region, which makes sense because these transitions all end at the ground state (n = 1), which means they involve relatively large energy changes. As we move to higher lines in the series (from the first to the second to the third, etc.), the wavelength increases (the photon energy decreases) because the energy difference between successive levels decreases. The third line at 97.2 nm is still well into the UV (UV extends from about 10 nm to 400 nm), though it’s longer than the first line (121 nm) and second line (103 nm). The series limit (as $n_i \to \infty$ ) approaches 91.2 nm, which represents the minimum energy needed to ionize hydrogen from its ground state.
Look up the values of the quantities in ${a}_{\text{B}}=\frac{ h^{2} }{ 4\pi^{2}{m}_{e} k q_{e}^{2} }$ , and verify that the Bohr radius ${a}_{\text{B}}$ is $0.529 \times 10^{-10} \text{m}$.
Strategy
We’ll substitute the known values of fundamental constants into the equation $a_{\text{B}} = \frac{h^2}{4\pi^2 m_e k q_e^2}$ and verify that the result is $0.529 \times 10^{-10} \text{ m}$ . The constants we need are:
Solution
Substituting these values into the Bohr radius equation:
This verifies that the Bohr radius is $0.529 \times 10^{-10} \text{ m}$ or 0.529 Å (angstroms).
Discussion
The Bohr radius represents the most probable distance of the electron from the nucleus in the ground state of the hydrogen atom. This fundamental length scale emerges naturally from the combination of quantum mechanics (Planck’s constant $h$ ), the properties of the electron (its mass $m_e$ and charge $q_e$ ), and the electromagnetic force (Coulomb’s constant $k$ ). The fact that this theoretical prediction matches experimental measurements of the hydrogen atom’s size was one of the great triumphs of Bohr’s theory. The Bohr radius serves as a natural unit for expressing atomic distances—for example, in hydrogen-like atoms, the radius of the n-th orbit is $r_n = n^2 a_{\text{B}}/Z$ . The small value (about 0.05 nm or half an angstrom) explains why atoms are invisible to the naked eye and why we need powerful microscopes or indirect methods to study atomic structure. It’s worth noting that in modern quantum mechanics, we no longer think of the electron as orbiting at a fixed radius; instead, the Bohr radius represents the peak of the radial probability distribution for finding the electron in the ground state.
Verify that the ground state energy ${E}_{0}$ is 13.6 eV by using ${E}_{0}=\frac{ {2\pi }^{2}{q}_{e}^{4}{m}_{e}{k}^{2}}{ {h}^{2}}\text{.}$
Strategy
We’ll substitute the known fundamental constants into the given equation and calculate the result. The constants we need are:
Solution
Substituting these values into the equation:
Working through the calculation step by step:
Converting to electron volts (dividing by $q_e = 1.602 \times 10^{-19} \text{ J/eV}$ ):
This verifies that the ground state energy of hydrogen is indeed 13.6 eV.
Discussion
This result is fundamental to atomic physics. The ground state energy of –13.6 eV (negative because it represents a bound state) means that 13.6 eV of energy is required to ionize a hydrogen atom from its ground state. This value emerges directly from the fundamental constants of nature—the electron’s charge and mass, Planck’s constant, and Coulomb’s constant. The fact that Bohr could derive this experimentally known value from first principles was a major triumph of his theory and provided strong support for the quantum model of the atom. The energy levels for excited states are given by $E_n = E_0/n^2$ , so all hydrogen energy levels can be calculated from this single fundamental value.
If a hydrogen atom has its electron in the $n=4$ state, how much energy in eV is needed to ionize it?
Strategy
The energy of an electron in the n-th energy level of hydrogen is given by $E_n = -\frac{13.6 \text{ eV}}{n^2}$ . To ionize the atom from the $n = 4$ state means bringing the electron from $E_4$ to zero energy (the ionization threshold). The ionization energy is the magnitude of $E_4$ .
Solution
For $n = 4$ :
The ionization energy is:
Therefore, 0.850 eV of energy is needed to ionize a hydrogen atom from the $n = 4$ state.
Discussion
This energy is much less than the 13.6 eV needed to ionize hydrogen from its ground state ( $n = 1$ ). The higher the energy level, the less tightly the electron is bound to the nucleus, and therefore the less energy is required to ionize it. This makes physical sense because the electron in the $n = 4$ state is farther from the nucleus (the orbital radius increases as $n^2$ ) and experiences a weaker attractive force. The ionization energy decreases as $1/n^2$ : for $n = 2$ it’s 3.4 eV, for $n = 3$ it’s 1.51 eV, and for $n = 4$ it’s 0.850 eV. As $n \to \infty$ , the ionization energy approaches zero, meaning the electron is barely bound. This is why highly excited atoms (high $n$ ) are easily ionized by collisions, electric fields, or photons, and why such “Rydberg atoms” are important in studying quantum-to-classical transitions and in applications like quantum computing.
A hydrogen atom in an excited state can be ionized with less energy than when it is in its ground state. What is $n$ for a hydrogen atom if 0.850 eV of energy can ionize it?
Strategy
| The energy of an electron in the $n$-th energy level of hydrogen is given by $E_n = -\frac{13.6 \text{ eV}}{n^2}$ . The energy is negative because the electron is in a bound state. To ionize the atom from level $n$ , we must provide enough energy to bring the electron from $E_n$ to zero energy (the ionization threshold). Therefore, the ionization energy is $$ | E_n | = \frac{13.6 \text{ eV}}{n^2} $. We can solve for$ n $$ . |
Solution
Setting the ionization energy equal to 0.850 eV:
Solving for $n^2$ :
Therefore, the hydrogen atom is in the n = 4 energy level.
Discussion
This result makes physical sense. The higher the energy level (larger $n$ ), the less tightly bound the electron is to the nucleus, and therefore the less energy is required to ionize it. For the ground state (n = 1), we need 13.6 eV to ionize hydrogen. For n = 2, we need 13.6/4 = 3.4 eV. For n = 3, we need 13.6/9 = 1.51 eV. And for n = 4, we need 13.6/16 = 0.850 eV, which matches the given ionization energy. As $n$ approaches infinity, the ionization energy approaches zero, which makes sense because the electron is already nearly free at very high quantum numbers. This problem demonstrates why excited atoms are more easily ionized than ground-state atoms—a fact that’s important in understanding phenomena like flame tests, gas discharge tubes, and ionized gases in stars.
Find the radius of a hydrogen atom in the $n=2$ state according to Bohr’s theory.
Strategy
According to Bohr’s theory, the radius of the n-th orbit in hydrogen is given by $r_n = n^2 a_{\text{B}}$ , where $a_{\text{B}} = 0.529 \times 10^{-10} \text{ m}$ is the Bohr radius. For the $n = 2$ state, we simply substitute $n = 2$ into this formula.
Solution
Using $r_n = n^2 a_{\text{B}}$ with $n = 2$ :
The radius of the $n = 2$ state is $2.12 \times 10^{-10} \text{ m}$ or 2.12 Å.
Discussion
The $n = 2$ state has a radius exactly 4 times larger than the ground state ( $n = 1$ ), demonstrating the $n^2$ scaling of orbital radius in Bohr’s model. This is the first excited state of hydrogen. When an atom transitions from $n = 2$ to $n = 1$ , it emits a photon at 121.6 nm (Lyman-alpha), the first line of the Lyman series. In modern quantum mechanics, we interpret this radius as the location of the peak in the radial probability distribution for the 2s orbital, or the average radius for 2p orbitals. The larger radius of excited states explains why excited atoms are physically bigger and more easily perturbed by external fields than ground-state atoms. The $n^2$ scaling also explains why very high-n “Rydberg atoms” can be macroscopically large—for example, atoms with $n = 100$ would have radii of about 0.5 micrometers, large enough to be affected by room-temperature thermal radiation.
Show that $\left(13.6 \text{eV}\right)/hc= 1.097 \times 10^{7} \text{m}=R$ (Rydberg’s constant), as discussed in the text.
Strategy
We need to calculate $\frac{13.6 \text{ eV}}{hc}$ and show it equals the Rydberg constant $R = 1.097 \times 10^7 \text{ m}^{-1}$ . We’ll use the convenient relationship $hc = 1240 \text{ eV·nm}$ to simplify the calculation.
Solution
Using the relationship $hc = 1240 \text{ eV·nm}$ :
Converting to m⁻¹ (since 1 nm = 10⁻⁹ m, so 1 nm⁻¹ = 10⁹ m⁻¹):
This is exactly equal to the Rydberg constant $R = 1.097 \times 10^7 \text{ m}^{-1}$ .
Discussion
This result beautifully demonstrates the connection between Bohr’s atomic theory and the empirically discovered Rydberg formula. The Rydberg constant, originally determined from experimental spectroscopic data, can now be understood as arising from the fundamental ground-state binding energy of hydrogen (13.6 eV) divided by $hc$ . This relationship appears in the formula for the wavelengths of spectral lines:
The fact that Bohr’s theory could not only explain the existence of discrete spectral lines but also derive the previously mysterious Rydberg constant from first principles was one of the theory’s greatest triumphs. It showed that quantum mechanics could make precise quantitative predictions, not just qualitative explanations, and established the validity of the quantum approach to atomic structure.
What is the smallest-wavelength line in the Balmer series? Is it in the visible part of the spectrum?
Strategy
The Balmer series corresponds to transitions ending at $n_f = 2$ . The smallest wavelength (highest energy) occurs when the initial state has the highest possible $n_i$ , which approaches infinity. We’ll use the Rydberg formula with $n_f = 2$ and $n_i \to \infty$ to find the series limit.
Solution
Using the Rydberg formula:
For the series limit, $n_f = 2$ and $n_i \to \infty$ , so $\frac{1}{n_i^2} \to 0$ :
The smallest wavelength in the Balmer series is 365 nm, which is in the ultraviolet region (just below the 400 nm threshold for visible light).
Discussion
The Balmer series limit at 365 nm represents the minimum energy photon that can ionize a hydrogen atom from the $n = 2$ state. All transitions in the Balmer series that we observe have wavelengths longer than 365 nm. The first four lines of the Balmer series are in the visible region (H-alpha at 656 nm is red, H-beta at 486 nm is blue-green, H-gamma at 434 nm is violet, and H-delta at 410 nm is deep violet), but as we move to higher transitions (higher initial $n$ values), the wavelengths get shorter and eventually cross into the UV at 365 nm. This series limit is important historically—Balmer discovered the empirical formula for hydrogen spectral lines by studying these visible lines, and his work laid the foundation for Bohr’s quantum theory of the atom. The fact that the series has a well-defined limit (rather than continuing indefinitely) was one of the key clues that atomic energy levels are quantized.
Show that the entire Paschen series is in the infrared part of the spectrum. To do this, you only need to calculate the shortest wavelength in the series.
Strategy
The Paschen series corresponds to transitions that end at $n_{\text{f}} = 3$ . The shortest wavelength (highest energy transition) in any series occurs when $n_i \to \infty$ , because this represents the largest possible energy difference for that series. If we can show that even this shortest wavelength is in the infrared (λ > 700 nm), then all other lines in the Paschen series must also be in the infrared, since they have longer wavelengths. We’ll use the Rydberg formula:
Solution
For the Paschen series, $n_{\text{f}} = 3$ . As $n_i \to \infty$ , the term $\frac{1}{n_i^2} \to 0$ , so:
Substituting $R = 1.097 \times 10^7 \text{ m}^{-1}$ :
Since the infrared region begins at wavelengths greater than about 700 nm, and the shortest wavelength in the Paschen series is 820 nm, the entire Paschen series lies in the infrared region.
Discussion
This result demonstrates why the Paschen series was discovered later than the Balmer series—it’s not visible to the human eye. The visible spectrum extends from approximately 400 nm (violet) to 700 nm (red), while infrared radiation has wavelengths longer than 700 nm. Since 820 nm is the shortest wavelength in the Paschen series, all other lines in the series (corresponding to transitions from $n_i = 4, 5, 6, ...$ to $n_f = 3$ ) must have even longer wavelengths and therefore are also in the infrared. For example, the first line of the Paschen series (from $n_i = 4$ to $n_f = 3$ ) has a wavelength of 1875 nm, which is well into the infrared. This series is important in astronomy for studying stellar atmospheres and in understanding the complete hydrogen spectrum.
Do the Balmer and Lyman series overlap? To answer this, calculate the shortest-wavelength Balmer line and the longest-wavelength Lyman line.
Strategy
To determine if the Balmer and Lyman series overlap, we need to compare the shortest wavelength of the Balmer series (its series limit) with the longest wavelength of the Lyman series (its first line). If the Balmer minimum is shorter than or equal to the Lyman maximum, the series would overlap. We’ll calculate both wavelengths using the Rydberg formula.
Solution
Shortest Balmer wavelength (series limit, $n_f = 2$, $n_i \to \infty$ ):
Longest Lyman wavelength (first line, $n_f = 1$, $n_i = 2$ ):
Since 365 nm > 122 nm, the Balmer and Lyman series do not overlap.
Discussion
The Balmer series spans from 365 nm (series limit) to 656 nm (H-alpha line), while the Lyman series spans from 91.2 nm (series limit) to 122 nm (Lyman-alpha). There’s a clear gap of 243 nm (from 122 nm to 365 nm) between these two series. This gap is significant—it’s nearly twice the width of the Lyman series itself. This separation makes sense because the Balmer series involves transitions to $n = 2$ while Lyman involves transitions to $n = 1$ , and the energy difference between these final states is substantial (10.2 eV). The non-overlapping nature of hydrogen’s spectral series made it easier for early spectroscopists to identify and categorize the different series. In contrast, for heavier hydrogen-like ions with larger $Z$ values, the energy levels scale as $Z^2$ , which can cause different series to overlap in wavelength even though they correspond to different transitions.
(a) Which line in the Balmer series is the first one in the UV part of the spectrum?
(b) How many Balmer series lines are in the visible part of the spectrum?
(c) How many are in the UV?
Strategy
The Balmer series corresponds to transitions ending at $n_{\text{f}} = 2$ , with initial states $n_i = 3, 4, 5, 6, ...$ . The visible spectrum extends from approximately 400 nm (violet) to 700 nm (red), while ultraviolet (UV) radiation has wavelengths shorter than 400 nm. We’ll calculate the wavelength for each Balmer line using the Rydberg formula until we find the transition into the UV region. Since wavelength decreases as $n_i$ increases, we need to find which value of $n_i$ produces the first wavelength below 400 nm.
Solution
For the Balmer series, $n_{\text{f}} = 2$ . Let’s calculate wavelengths for successive values of $n_i$ :
For $n_i = 3$ (H-alpha line):
For $n_i = 4$ (H-beta line):
For $n_i = 5$ (H-gamma line):
For $n_i = 6$ (H-delta line):
For $n_i = 7$ (H-epsilon line):
(a) The line corresponding to the transition from $n_i = 7$ to $n_f = 2$ (H-epsilon line) is the first one in the UV part of the spectrum, with $\lambda = 397 \text{ nm}$ .
(b) Four Balmer series lines are in the visible part of the spectrum (corresponding to $n_i = 3, 4, 5, 6$ ).
(c) Infinitely many Balmer lines are in the UV. As $n_i$ increases from 7 to infinity, all wavelengths become progressively shorter, remaining in the UV and approaching the series limit of 365 nm.
Discussion
The Balmer series is special because it’s the only hydrogen spectral series with lines in the visible region—this is why it was discovered first and studied most extensively in early spectroscopy. The four visible lines (H-alpha at 656 nm appearing red, H-beta at 486 nm appearing blue-green, H-gamma at 434 nm appearing violet, and H-delta at 410 nm appearing violet) can be seen with the naked eye in hydrogen discharge tubes and in astronomical observations of nebulae and stars. The fact that only four lines are visible while infinitely many are in the UV illustrates how the energy levels become more closely spaced as $n$ increases. The series limit (as $n_i \to \infty$ ) is at 365 nm, representing the energy needed to ionize hydrogen from the $n = 2$ state. This wavelength marks the shortest possible Balmer line and separates discrete spectral lines from the continuum that appears beyond the series limit.
A wavelength of $4.653 \text{μm}$ is observed in a hydrogen spectrum for a transition that ends in the ${n}_{\text{f}}=5$ level. What was ${n}_{\text{i}}$ for the initial level of the electron?
Strategy
We’re given the wavelength $\lambda = 4.653 \text{ μm}$ and the final level $n_f = 5$ . We need to find the initial level $n_i$ . Using the Rydberg formula, we can solve for $n_i$ : $\frac{1}{\lambda} = R \left( \frac{1}{n_f^2} - \frac{1}{n_i^2} \right)$ .
Solution
First, convert the wavelength to meters:
Using the Rydberg formula:
The electron was initially in the n = 7 level.
Discussion
This transition from $n = 7$ to $n = 5$ produces infrared radiation at 4.653 μm, which is well beyond the visible range. This is part of what we might call an extended Pfund series (transitions ending at $n = 5$ ). The Pfund series includes several lines in the mid-infrared, all invisible to the human eye but detectable with infrared spectrometers. The long wavelength (low energy) of this transition makes sense because both initial and final states are highly excited, so the energy difference is relatively small. Transitions between high-lying states are important in astrophysics for studying the conditions in stellar atmospheres and interstellar gas clouds, where hydrogen atoms can be excited to these high levels by the intense radiation fields. These infrared hydrogen lines are also used to study the kinematics of gas in galaxies through Doppler shifts.
A singly ionized helium ion has only one electron and is denoted ${\text{He}}^{+}$ . What is the ion’s radius in the ground state compared to the Bohr radius of hydrogen atom?
Strategy
A singly ionized helium ion ( $\text{He}^+$ ) is a hydrogen-like atom with one electron orbiting a nucleus containing two protons (so $Z = 2$ ). We can use the general formula for the radius of hydrogen-like atoms:
where $a_{\text{B}} = 0.529 \times 10^{-10} \text{ m}$ is the Bohr radius. For the ground state, $n = 1$ .
Solution
For $\text{He}^+$ in the ground state, we have $n = 1$ and $Z = 2$ :
Therefore, the radius of $\text{He}^+$ in its ground state is half the Bohr radius of hydrogen, or:
Discussion
This result makes physical sense: the helium nucleus has twice the positive charge of the hydrogen nucleus, so it exerts a stronger attractive force on the electron. This stronger force pulls the electron into a tighter orbit, reducing the orbital radius by a factor of 2. More generally, for any hydrogen-like ion in the ground state, the radius is inversely proportional to the atomic number: $r_1 = a_{\text{B}}/Z$ . This means that highly ionized atoms (large $Z$ ) have very small radii. For example, $\text{Li}^{2+}$ (lithium with two electrons removed, $Z = 3$ ) would have a ground-state radius of $a_{\text{B}}/3$ . The same principle applies to energy levels: the binding energy of $\text{He}^+$ is proportional to $Z^2 = 4$ , making it four times more tightly bound than hydrogen (54.4 eV compared to 13.6 eV for hydrogen).
A beryllium ion with a single electron (denoted ${\text{Be}}^{3+}$ ) is in an excited state with radius the same as that of the ground state of hydrogen.
(a) What is $n$ for the ${\text{Be}}^{3+}$ ion?
(b) How much energy in eV is needed to ionize the ion from this excited state?
Strategy
For hydrogen-like ions (single electron), the orbital radius is given by $r_n = \frac{n^2}{Z} a_{\text{B}}$ and the energy is $E_n = -\frac{Z^2}{n^2}(13.6 \text{ eV})$ , where $Z$ is the atomic number. For Be³⁺, $Z = 4$ . (a) We’ll find which $n$ gives the same radius as hydrogen’s ground state ( $r = a_{\text{B}}$ ). (b) We’ll calculate the ionization energy from that state.
Solution
(a) The ground state of hydrogen has radius $r_1 = a_{\text{B}}$ . For Be³⁺ with $Z = 4$ :
Setting this equal to $a_{\text{B}}$ :
The Be³⁺ ion is in the n = 2 state.
(b) The energy of the $n = 2$ state for Be³⁺ is:
The ionization energy is the energy needed to bring the electron from $E_2 = -54.4 \text{ eV}$ to $E = 0$ :
The ionization energy is 54.4 eV.
Discussion
This problem beautifully illustrates how the $Z^2$ scaling affects both orbital radii and energies in hydrogen-like ions. The Be³⁺ ion has $Z = 4$ , so its orbitals are compressed by a factor of $Z = 4$ (radius scales as $1/Z$ ) compared to hydrogen at the same $n$ . To match hydrogen’s ground state radius, the Be³⁺ electron must be in $n = 2$ , which is its first excited state. However, despite having the same radius as hydrogen’s ground state, the Be³⁺ ion in $n = 2$ is much more tightly bound—it requires 54.4 eV to ionize, exactly 4 times the 13.6 eV needed for hydrogen’s ground state (energy scales as $Z^2$ ). This shows that nuclear charge dominates the binding energy. Be³⁺ is created in very hot environments like stellar interiors or in high-energy laboratory plasmas. The high ionization energy of 54.4 eV (even from an excited state!) means that only very energetic photons (in the soft X-ray region, around 23 nm wavelength) or very energetic collisions can ionize it. For comparison, the ground state ($n = 1$ ) of Be³⁺ would require 217.6 eV to ionize, placing it in the extreme UV to soft X-ray spectral region.
Atoms can be ionized by thermal collisions, such as at the high temperatures found in the solar corona. One such ion is ${C}^{+5}$ , a carbon atom with only a single electron.
(a) By what factor are the energies of its hydrogen-like levels greater than those of hydrogen?
(b) What is the wavelength of the first line in this ion’s Paschen series?
(c) What type of EM radiation is this?
Strategy
The ion $\text{C}^{+5}$ is a carbon atom with 5 electrons removed, leaving only one electron. Since carbon has 6 protons, $Z = 6$ for this hydrogen-like ion. For hydrogen-like atoms, the energy levels are given by:
where $E_0 = 13.6 \text{ eV}$ is the ground-state energy of hydrogen. The energy of a photon emitted during a transition is $\Delta E = E_i - E_f$ , which can be related to wavelength using $E = hc/\lambda$ .
Solution
(a) The energy levels of $\text{C}^{+5}$ are greater than those of hydrogen by a factor of $Z^2$ :
The energies are 36 times greater than those of hydrogen.
(b) For the Paschen series, $n_{\text{f}} = 3$ . The first line corresponds to a transition from $n_i = 4$ to $n_f = 3$ . The energy of the emitted photon is:
Converting to wavelength using $\lambda = \frac{hc}{\Delta E}$ and $hc = 1240 \text{ eV·nm}$ :
(c) A wavelength of 52.1 nm is in the ultraviolet (UV) region of the electromagnetic spectrum, specifically in the extreme ultraviolet (EUV) or far-UV range.
Discussion
This problem illustrates how hydrogen-like ions with high atomic numbers produce much higher energy transitions than hydrogen itself. The factor of 36 increase in energy (due to $Z^2 = 36$ ) means that what would be an infrared transition in hydrogen (the Paschen series) becomes an extreme ultraviolet transition in $\text{C}^{+5}$ . For comparison, the first line of the Paschen series in hydrogen has a wavelength of 1875 nm (infrared), while the same transition in $\text{C}^{+5}$ has a wavelength 36 times shorter: 52.1 nm (extreme UV). This is exactly what we expect since $\lambda \propto 1/(\Delta E) \propto 1/Z^2$ . Such highly ionized carbon atoms are found in the solar corona and other high-temperature astrophysical plasmas where temperatures exceed one million Kelvin. The extreme UV radiation from these ions is important for understanding solar physics and space weather, though it is absorbed by Earth’s atmosphere and must be observed from space-based telescopes.
Verify Equations ${r}_{n}=\frac{ {n}^{2}}{Z}{a}_{\text{B}}$ and ${a}_{B}=\frac{ {h}^{2}}{ {4\pi }^{2}{m}_{e} k q_{e}^{2}}= 0.529 \times 10^{-10} \text{m}$ using the approach stated in the text. That is, equate the Coulomb and centripetal forces and then insert an expression for velocity from the condition for angular momentum quantization.
Strategy
We’ll follow Bohr’s approach by: (1) equating the electrostatic Coulomb force to the centripetal force needed to keep the electron in circular orbit, and (2) using the quantization condition for angular momentum $L = m_e v r_n = n\frac{h}{2\pi}$ to eliminate the velocity. This will yield expressions for $r_n$ in terms of fundamental constants and quantum number $n$ .
Solution
Starting with the force balance equation, the Coulomb attractive force equals the centripetal force:
Multiplying both sides by $r_n$ and rearranging:
From Bohr’s quantization condition:
Solving for $v$ :
Substituting this expression for $v$ into our equation for $r_n$ :
Simplifying:
Defining the Bohr radius as:
We obtain the general formula:
This verifies both equations.
Discussion
This derivation is at the heart of Bohr’s atomic model and represents a brilliant synthesis of classical and quantum ideas. The force balance equation (Coulomb force = centripetal force) is pure classical mechanics, while the quantization condition $L = n\frac{h}{2\pi}$ introduces quantum mechanics. The resulting formula $r_n = \frac{n^2}{Z} a_{\text{B}}$ has profound implications: (1) Orbital radii are quantized and scale as $n^2$ , explaining why higher energy states are much larger; (2) For hydrogen-like ions, the radius scales as $1/Z$ , meaning higher nuclear charge squeezes the electron orbits inward; (3) The Bohr radius $a_{\text{B}}$ emerges as a natural length scale built from fundamental constants, representing the ground state size of hydrogen. At the time (1913), this theory successfully explained the Rydberg formula for hydrogen spectral lines and predicted the radii of excited states. Although superseded by full quantum mechanics, Bohr’s model remains pedagogically valuable and gives surprisingly accurate results for hydrogen and hydrogen-like ions. The approach also introduced the revolutionary idea that quantum numbers (like $n$ ) determine discrete atomic properties—a concept that became central to all of quantum physics.
The wavelength of the four Balmer series lines for hydrogen are found to be 410.3, 434.2, 486.3, and 656.5 nm. What average percentage difference is found between these wavelength numbers and those predicted by $\frac{1}{\lambda }=R\left(\frac{1}{ {n}_{\text{f}}^{2}}-\frac{1}{ {n}_{\text{i}}^{2}}\right)$ ? It is amazing how well a simple formula (disconnected originally from theory) could duplicate this phenomenon.
Strategy
The Balmer series has $n_{\text{f}} = 2$ , and the four lines correspond to transitions from $n_i = 6, 5, 4, 3$ (in order of the given experimental wavelengths from shortest to longest). We’ll calculate the theoretical wavelength for each transition using the Rydberg formula, compare with the experimental values, calculate the percentage difference for each line, and then find the average.
Solution
For the Balmer series with $R = 1.097 \times 10^7 \text{ m}^{-1}$ and $n_f = 2$ :
Line 1: $n_i = 6$ (experimental: 410.3 nm)
Line 2: $n_i = 5$ (experimental: 434.2 nm)
Line 3: $n_i = 4$ (experimental: 486.3 nm)
Line 4: $n_i = 3$ (experimental: 656.5 nm)
Average percentage difference:
The average percentage difference is approximately 0.03% or about 0.3 parts per thousand.
Discussion
This remarkably small average percentage difference (only about 0.03%) demonstrates the extraordinary accuracy of the Rydberg formula in predicting hydrogen spectral lines. The fact that such a simple empirical formula—originally developed by Balmer and Rydberg before the development of quantum mechanics—could predict wavelengths to within a few hundredths of a percent is truly remarkable. This agreement provided strong evidence that there was a fundamental physical principle underlying atomic spectra, even before Bohr developed his quantum theory of the hydrogen atom. The small discrepancies that do exist (all less than 0.05%) can be attributed to several factors: experimental uncertainties in measuring wavelengths, the finite mass of the nucleus (which we assumed to be infinite in the simple Bohr model), and relativistic corrections for the electron’s motion. When Bohr showed that his quantum theory could derive the Rydberg formula from first principles and explain why it worked so well, it was a watershed moment in physics that validated the quantum approach to atomic structure.