
When light strikes materials, it can eject electrons from them. This is called the photoelectric effect, meaning that light (photo) produces electricity. One common use of the photoelectric effect is in light meters, such as those that adjust the automatic iris on various types of cameras. In a similar way, another use is in solar cells, as you probably have in your calculator or have seen on a roof top or a roadside sign. These make use of the photoelectric effect to convert light into electricity for running different devices.

This effect has been known for more than a century and can be studied using a device such as that shown in [Figure 1]. This figure shows an evacuated tube with a metal plate and a collector wire that are connected by a variable voltage source, with the collector more negative than the plate. When light (or other EM radiation) strikes the plate in the evacuated tube, it may eject electrons. If the electrons have energy in electron volts (eV) greater than the potential difference between the plate and the wire in volts, some electrons will be collected on the wire. Since the electron energy in eV is $qV$ , where $q$ is the electron charge and $V$ is the potential difference, the electron energy can be measured by adjusting the retarding voltage between the wire and the plate. The voltage that stops the electrons from reaching the wire equals the energy in eV. For example, if $-3.00 \text{V}$ barely stops the electrons, their energy is 3.00 eV. The number of electrons ejected can be determined by measuring the current between the wire and plate. The more light, the more electrons; a little circuitry allows this device to be used as a light meter.
What is really important about the photoelectric effect is what Albert Einstein deduced from it. Einstein realized that there were several characteristics of the photoelectric effect that could be explained only if EM radiation is itself quantized: the apparently continuous stream of energy in an EM wave is actually composed of energy quanta called photons. In his explanation of the photoelectric effect, Einstein defined a quantized unit or quantum of EM energy, which we now call a photon, with an energy proportional to the frequency of EM radiation. In equation form, the photon energy is
where $E$ is the energy of a photon of frequency $f$ and $h$ is Planck’s constant. This revolutionary idea looks similar to Planck’s quantization of energy states in blackbody oscillators, but it is quite different. It is the quantization of EM radiation itself. EM waves are composed of photons and are not continuous smooth waves as described in previous chapters on optics. Their energy is absorbed and emitted in lumps, not continuously. This is exactly consistent with Planck’s quantization of energy levels in blackbody oscillators, since these oscillators increase and decrease their energy in steps of $hf$ by absorbing and emitting photons having $E= hf$ . We do not observe this with our eyes, because there are so many photons in common light sources that individual photons go unnoticed. (See [Figure 2].) The next section of the text (Photon Energies and the Electromagnetic Spectrum) is devoted to a discussion of photons and some of their characteristics and implications. For now, we will use the photon concept to explain the photoelectric effect, much as Einstein did.
The photoelectric effect has the properties discussed below. All these properties are consistent with the idea that individual photons of EM radiation are absorbed by individual electrons in a material, with the electron gaining the photon’s energy. Some of these properties are inconsistent with the idea that EM radiation is a simple wave. For simplicity, let us consider what happens with monochromatic EM radiation in which all photons have the same energy $hf$
.
If we vary the frequency of the EM radiation falling on a material, we find the following: For a given material, there is a threshold frequency ${f}_{0}$ for the EM radiation below which no electrons are ejected, regardless of intensity. Individual photons interact with individual electrons. Thus if the photon energy is too small to break an electron away, no electrons will be ejected. If EM radiation was a simple wave, sufficient energy could be obtained by increasing the intensity. 2. Once EM radiation falls on a material, electrons are ejected without delay. As soon as an individual photon of a sufficiently high frequency is absorbed by an individual electron, the electron is ejected. If the EM radiation were a simple wave, several minutes would be required for sufficient energy to be deposited to the metal surface to eject an electron. 3. The number of electrons ejected per unit time is proportional to the intensity of the EM radiation and to no other characteristic. High-intensity EM radiation consists of large numbers of photons per unit area, with all photons having the same characteristic energy $hf$ .
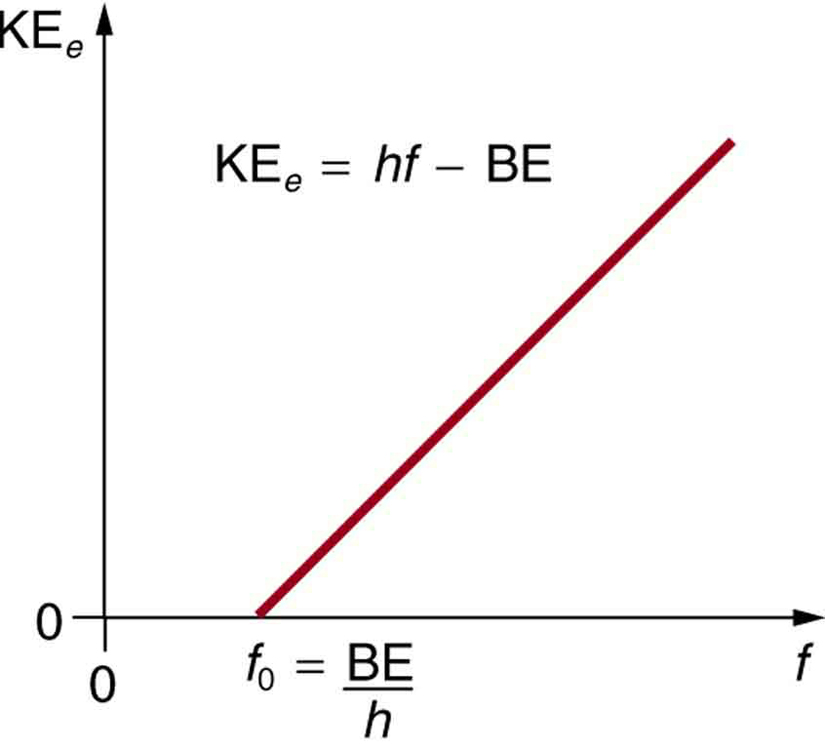
where ${\text{KE}}_{e}$ is the maximum kinetic energy of the ejected electron, $hf$ is the photon’s energy, and BE is the binding energy of the electron to the particular material. (BE is sometimes called the work function of the material.) This equation, due to Einstein in 1905, explains the properties of the photoelectric effect quantitatively. An individual photon of EM radiation (it does not come any other way) interacts with an individual electron, supplying enough energy, BE, to break it away, with the remainder going to kinetic energy. The binding energy is $\text{BE}=hf_{0}$ , where ${f}_{0}$ is the threshold frequency for the particular material. [Figure 3] shows a graph of maximum ${\text{KE}}\_{e}$
versus the frequency of incident EM radiation falling on a particular material.
Einstein’s idea that EM radiation is quantized was crucial to the beginnings of quantum mechanics. It is a far more general concept than its explanation of the photoelectric effect might imply. All EM radiation can also be modeled in the form of photons, and the characteristics of EM radiation are entirely consistent with this fact. (As we will see in the next section, many aspects of EM radiation, such as the hazards of ultraviolet (UV) radiation, can be explained _ only_ by photon properties.) More famous for modern relativity, Einstein planted an important seed for quantum mechanics in 1905, the same year he published his first paper on special relativity. His explanation of the photoelectric effect was the basis for the Nobel Prize awarded to him in 1921. Although his other contributions to theoretical physics were also noted in that award, special and general relativity were not fully recognized in spite of having been partially verified by experiment by 1921. Although hero-worshipped, this great man never received Nobel recognition for his most famous work—relativity.
(a) What is the energy in joules and electron volts of a photon of 420-nm violet light? (b) What is the maximum kinetic energy of electrons ejected from calcium by 420-nm violet light, given that the binding energy (or work function) of electrons for calcium metal is 2.71 eV?
Strategy
To solve part (a), note that the energy of a photon is given by $E= hf$ . For part (b), once the energy of the photon is calculated, it is a straightforward application of ${\text{KE}}_{e}= hf- \text{BE}$ to find the ejected electron’s maximum kinetic energy, since BE is given.
Solution for (a)
Photon energy is given by
Since we are given the wavelength rather than the frequency, we solve the familiar relationship $c=f\lambda$ for the frequency, yielding
Combining these two equations gives the useful relationship
Now substituting known values yields
Converting to eV, the energy of the photon is
Solution for (b)
Finding the kinetic energy of the ejected electron is now a simple application of the equation ${\text{KE}}_{e}= hf- \text{BE}$ . Substituting the photon energy and binding energy yields
Discussion
The energy of this 420-nm photon of violet light is a tiny fraction of a joule, and so it is no wonder that a single photon would be difficult for us to sense directly—humans are more attuned to energies on the order of joules. But looking at the energy in electron volts, we can see that this photon has enough energy to affect atoms and molecules. A DNA molecule can be broken with about 1 eV of energy, for example, and typical atomic and molecular energies are on the order of eV, so that the UV photon in this example could have biological effects. The ejected electron (called a photoelectron) has a rather low energy, and it would not travel far, except in a vacuum. The electron would be stopped by a retarding potential of but 0.26 eV. In fact, if the photon wavelength were longer and its energy less than 2.71 eV, then the formula would give a negative kinetic energy, an impossibility. This simply means that the 420-nm photons with their 2.96-eV energy are not much above the frequency threshold. You can show for yourself that the threshold wavelength is 459 nm (blue light). This means that if calcium metal is used in a light meter, the meter will be insensitive to wavelengths longer than those of blue light. Such a light meter would be completely insensitive to red light, for example.
See how light knocks electrons off a metal target, and recreate the experiment that spawned the field of quantum mechanics.
Is visible light the only type of EM radiation that can cause the photoelectric effect?
Which aspects of the photoelectric effect cannot be explained without photons? Which can be explained without photons? Are the latter inconsistent with the existence of photons?
Is the photoelectric effect a direct consequence of the wave character of EM radiation or of the particle character of EM radiation? Explain briefly.
Insulators (nonmetals) have a higher BE than metals, and it is more difficult for photons to eject electrons from insulators. Discuss how this relates to the free charges in metals that make them good conductors.
If you pick up and shake a piece of metal that has electrons in it free to move as a current, no electrons fall out. Yet if you heat the metal, electrons can be boiled off. Explain both of these facts as they relate to the amount and distribution of energy involved with shaking the object as compared with heating it.
What is the longest-wavelength EM radiation that can eject a photoelectron from silver, given that the binding energy is 4.73 eV? Is this in the visible range?
Strategy
The longest wavelength corresponds to the minimum photon energy needed to eject an electron, which equals the binding energy. We use $E = hf = \frac{hc}{\lambda}$ with $E = \text{BE}$.
Solution
At the threshold for photoemission, the photon energy equals the binding energy:
Solving for wavelength:
Using $hc = 1240 \text{ eV·nm}$:
Discussion
This wavelength of 262 nm is in the ultraviolet range, well below the visible spectrum which extends from approximately 380 nm (violet) to 760 nm (red). Silver requires UV radiation for the photoelectric effect because its binding energy (4.73 eV) is higher than the photon energies of visible light (1.63-3.26 eV). Photons with wavelengths shorter than 262 nm (higher energy) can eject electrons from silver, while longer wavelengths cannot. This high binding energy makes silver less suitable for photoelectric devices that operate with visible light but useful for UV detection applications.
263 nm, no this is not visible (UV radiation)
Find the longest-wavelength photon that can eject an electron from potassium, given that the binding energy is 2.24 eV. Is this visible EM radiation?
Strategy
The longest wavelength corresponds to the minimum photon energy needed to eject an electron, which equals the binding energy. We use $E = hf = \frac{hc}{\lambda}$ with $E = \text{BE}$.
Solution
At the threshold for photoemission, the photon energy equals the binding energy:
Solving for wavelength:
Using $hc = 1240 \text{ eV·nm}$:
Discussion
This wavelength of 554 nm is in the visible range, specifically in the green-yellow part of the spectrum. Visible light ranges from approximately 380 nm (violet) to 760 nm (red), so this threshold wavelength is indeed visible. Photons with wavelengths shorter than 554 nm (higher energy) can eject electrons from potassium, while longer wavelengths cannot. This makes potassium useful in photoelectric devices sensitive to visible light.
554 nm, yes this is visible (green-yellow light)
What is the binding energy in eV of electrons in magnesium, if the longest-wavelength photon that can eject electrons is 337 nm?
Strategy
The longest wavelength that can eject electrons corresponds to photons with energy equal to the binding energy. We use $\text{BE} = hf = \frac{hc}{\lambda}$.
Solution
The binding energy equals the photon energy at threshold:
Using $hc = 1240 \text{ eV·nm}$:
Discussion
Magnesium has a binding energy of 3.68 eV, which falls between that of potassium (2.24 eV) and silver (4.73 eV). The threshold wavelength of 337 nm is in the ultraviolet range, just below the visible spectrum. This means magnesium requires UV light to exhibit the photoelectric effect and will not respond to visible light. This binding energy reflects the strength with which electrons are held in magnesium’s outer shell and is important for applications in UV photodetectors and understanding magnesium’s chemical properties.
3.69 eV
Calculate the binding energy in eV of electrons in aluminum, if the longest-wavelength photon that can eject them is 304 nm.
Strategy
The longest wavelength that can eject electrons corresponds to photons with energy equal to the binding energy. We use $\text{BE} = hf = \frac{hc}{\lambda}$.
Solution
The binding energy equals the photon energy at threshold:
Using $hc = 1240 \text{ eV·nm}$:
Discussion
Aluminum has a binding energy of 4.08 eV, which is higher than that of potassium (2.24 eV) and sodium (2.28 eV) but lower than silver (4.73 eV). The threshold wavelength of 304 nm is in the ultraviolet range, just below the visible spectrum. This means aluminum surfaces require UV light or shorter wavelengths to exhibit the photoelectric effect, making aluminum less sensitive than alkali metals for visible-light photoelectric applications.
4.08 eV
What is the maximum kinetic energy in eV of electrons ejected from sodium metal by 450-nm EM radiation, given that the binding energy is 2.28 eV?
Strategy
Use the photoelectric equation ${\text{KE}}_e = hf - \text{BE}$, where the photon energy is $hf = \frac{hc}{\lambda}$. Calculate the photon energy first, then subtract the binding energy.
Solution
First, calculate the photon energy using $hc = 1240 \text{ eV·nm}$:
Now apply the photoelectric equation:
Discussion
The 450-nm blue light photons have just barely enough energy (2.76 eV) to overcome sodium’s binding energy (2.28 eV) and eject electrons with a small kinetic energy of 0.48 eV. This demonstrates that sodium is sensitive to visible light, making it useful for photoelectric applications. The small kinetic energy means these photoelectrons won’t travel far and can be stopped by a retarding potential of only 0.48 V.
0.483 eV
UV radiation having a wavelength of 120 nm falls on gold metal, to which electrons are bound by 4.82 eV. What is the maximum kinetic energy of the ejected photoelectrons?
Strategy
We use the photoelectric equation ${\text{KE}}_e = hf - \text{BE}$, where the photon energy is $hf = \frac{hc}{\lambda}$. The maximum kinetic energy is the photon energy minus the binding energy.
Solution
First, calculate the photon energy using $hc = 1240 \text{ eV·nm}$:
Now apply the photoelectric equation:
Discussion
The UV photon has sufficient energy (10.33 eV) to overcome the binding energy of gold (4.82 eV) and still impart 5.51 eV of kinetic energy to the ejected electron. This is a relatively high kinetic energy for a photoelectron, more than twice the binding energy. Such energetic photoelectrons could travel significant distances in a vacuum before being stopped. Gold’s relatively high binding energy makes it less suitable for visible-light photoelectric applications but useful for UV detection.
5.51 eV
Violet light of wavelength 400 nm ejects electrons with a maximum kinetic energy of 0.860 eV from sodium metal. What is the binding energy of electrons to sodium metal?
Strategy
Use the photoelectric equation ${\text{KE}}_e = hf - \text{BE}$ and solve for the binding energy. First calculate the photon energy, then use the given kinetic energy to find BE.
Solution
Calculate the photon energy using $hc = 1240 \text{ eV·nm}$:
Rearrange the photoelectric equation to solve for binding energy:
Discussion
This value of 2.24 eV matches the known work function of sodium within experimental uncertainty. The 400-nm violet photons carry 3.10 eV of energy, of which 2.24 eV is needed to overcome the binding and 0.860 eV goes into the electron’s kinetic energy. This relatively low binding energy makes sodium highly sensitive to visible light, which is why sodium-based photocells are common in practical photoelectric devices. The measurement demonstrates how the photoelectric effect can be used to determine the work function of materials.
2.25 eV
UV radiation having a 300-nm wavelength falls on uranium metal, ejecting 0.500-eV electrons. What is the binding energy of electrons to uranium metal?
Strategy
We use the photoelectric equation ${\text{KE}}_e = hf - \text{BE}$ and solve for the binding energy. First calculate the photon energy, then use the given kinetic energy to find BE.
Solution
Calculate the photon energy using $hc = 1240 \text{ eV·nm}$:
Rearrange the photoelectric equation to solve for binding energy:
Discussion
Uranium metal has a binding energy of 3.63 eV for its outermost electrons. This value is intermediate between the alkali metals (around 2-2.3 eV) and more tightly bound metals like silver (4.73 eV). The 300-nm UV photon provides 4.13 eV of energy, which is sufficient to overcome the binding and give the electron 0.500 eV of kinetic energy. This binding energy indicates uranium requires UV radiation for photoelectric emission, as visible light photons lack sufficient energy.
3.63 eV
What is the wavelength of EM radiation that ejects 2.00-eV electrons from calcium metal, given that the binding energy is 2.71 eV? What type of EM radiation is this?
Strategy
The photon energy must equal the sum of binding energy and kinetic energy: $E_{\text{photon}} = \text{BE} + {\text{KE}}_e$. We then use $\lambda = \frac{hc}{E_{\text{photon}}}$ to find the wavelength.
Solution
The total photon energy needed is:
Now find the wavelength using $hc = 1240 \text{ eV·nm}$:
This wavelength is in the ultraviolet (UV) region of the electromagnetic spectrum.
Discussion
The 263-nm UV photons carry 4.71 eV of energy, with 2.71 eV used to overcome calcium’s binding energy and 2.00 eV transferred to the electron as kinetic energy. This is well into the UV range (below 400 nm), meaning calcium requires UV radiation to produce photoelectrons with this much kinetic energy. Visible light photons lack sufficient energy for this process. The relatively high photon energy requirement demonstrates why UV radiation is more effective at causing the photoelectric effect than visible light for most metals.
(a) 264 nm
(b) Ultraviolet
Find the wavelength of photons that eject 0.100-eV electrons from potassium, given that the binding energy is 2.24 eV. Are these photons visible?
Strategy
The photon energy must equal the sum of binding energy and kinetic energy: $E_{\text{photon}} = \text{BE} + {\text{KE}}_e$. We then use $\lambda = \frac{hc}{E_{\text{photon}}}$ to find the wavelength.
Solution
The total photon energy needed is:
Now find the wavelength using $hc = 1240 \text{ eV·nm}$:
Discussion
These photons have a wavelength of 530 nm, which is in the visible spectrum—specifically, green light. The visible spectrum ranges from about 380 nm (violet) to 760 nm (red), so 530 nm falls well within this range. Photons of this wavelength can eject electrons from potassium with a small kinetic energy of 0.100 eV, demonstrating that potassium is sensitive to visible light and is useful for photoelectric applications with visible radiation.
530 nm, yes these photons are visible (green light)
What is the maximum velocity of electrons ejected from a material by 80-nm photons, if they are bound to the material by 4.73 eV?
Strategy
First find the kinetic energy of the ejected electrons using ${\text{KE}}_e = hf - \text{BE}$, then use ${\text{KE}} = \frac{1}{2}mv^2$ to find velocity.
Solution
Calculate the photon energy using $hc = 1240 \text{ eV·nm}$:
Find the kinetic energy of ejected electrons:
Convert to joules: ${\text{KE}}_e = 10.77 \text{ eV} \times 1.60 \times 10^{-19} \text{ J/eV} = 1.72 \times 10^{-18} \text{ J}$
Use ${\text{KE}} = \frac{1}{2}mv^2$ to find velocity:
Discussion
The 80-nm UV photons carry significant energy (15.5 eV), producing fast-moving photoelectrons with velocities near 2 million m/s—about 0.6% of the speed of light. This is fast enough that we’re approaching the regime where relativistic effects begin to matter (though still well within the non-relativistic approximation). The high electron velocity demonstrates the substantial energy transfer possible with short-wavelength UV radiation, which is why UV photodetectors can produce strong electrical signals.
$1.95 \times 10^{6} \text{m/s}$
Photoelectrons from a material with a binding energy of 2.71 eV are ejected by 420-nm photons. Once ejected, how long does it take these electrons to travel 2.50 cm to a detection device?
Strategy
First, find the kinetic energy of the ejected electrons using ${\text{KE}}_e = hf - \text{BE}$. Then convert this to velocity using ${\text{KE}}_e = \frac{1}{2}mv^2$, assuming nonrelativistic speeds. Finally, use $t = \frac{d}{v}$ to find the time.
Solution
Calculate the photon energy:
Find the kinetic energy of ejected electrons:
Convert to joules:
Solve for velocity from ${\text{KE}}_e = \frac{1}{2}mv^2$:
Calculate the time to travel 2.50 cm:
Discussion
The photoelectrons travel at about 0.1% the speed of light (290 km/s), which is fast but well below relativistic speeds. They reach the detector in only 86.2 nanoseconds. This rapid transit time is important in photoelectric devices where quick response is essential. The relatively low kinetic energy (0.24 eV) results because the 420-nm photon energy (2.95 eV) is only slightly above the binding energy threshold (2.71 eV).
$8.62 \times 10^{-8} \text{ s}$ or 86.2 ns
A laser with a power output of 2.00 mW at a wavelength of 400 nm is projected onto calcium metal. (a) How many electrons per second are ejected? (b) What power is carried away by the electrons, given that the binding energy is 2.71 eV?
Strategy
(a) Find the photon energy, then divide the total power by energy per photon to get photons/second. Assuming each photon ejects one electron gives electrons/second. (b) Find the kinetic energy per electron using the photoelectric equation, then multiply by the electron ejection rate.
Solution for (a)
Calculate the photon energy using $hc = 1240 \text{ eV·nm}$:
Number of photons (and electrons) per second:
Solution for (b)
Kinetic energy per electron:
Power carried by electrons:
Discussion
The laser produces about 4 trillion electrons per second, but the electrons carry away only 0.251 mW—about 12.6% of the input power. The rest (1.75 mW, or 87.4%) goes into overcoming the binding energy and is converted to heat in the calcium metal. This illustrates the inefficiency of the photoelectric effect for power conversion: most of the photon energy is “wasted” overcoming the work function rather than creating useful kinetic energy in the photoelectrons.
(a) $4.02 \times 10^{15} \text{/s}$ (b) 0.256 mW
(a) Calculate the number of photoelectrons per second ejected from a 1.00-mm 2 area of sodium metal by 500-nm EM radiation having an intensity of $1.30 {\text{kW/m}}^{2}$ (the intensity of sunlight above the Earth’s atmosphere). (b) Given that the binding energy is 2.28 eV, what power is carried away by the electrons? (c) The electrons carry away less power than brought in by the photons. Where does the other power go? How can it be recovered?
Strategy
(a) Find the power incident on the area, then the energy per photon, and finally the number of photons (and electrons) per second. (b) Calculate the kinetic energy per electron and multiply by the rate. (c) Compare the input and output power to account for energy conservation.
Solution for (a)
Power incident on the area:
Energy per photon:
Number of photons (and electrons) per second:
Solution for (b)
Kinetic energy per electron:
Power carried by electrons:
Solution for (c)
Power used to overcome binding energy:
This energy goes into heating the sodium metal. It can be recovered as thermal energy or used to keep the metal at operating temperature.
Discussion
Only about 8% of the incident photon energy is carried away as kinetic energy of the electrons; the remaining 92% goes into overcoming the binding energy and heats the metal. This demonstrates why photoelectric devices can become warm during operation and why efficient cooling may be necessary for high-intensity applications.
(a) $3.27 \times 10^{15} \text{ electrons/s}$
(b) 0.105 W or 105 mW
(c) The remaining power (1.20 W) heats the sodium metal. It can be recovered as thermal energy or dissipated through cooling.
Unreasonable Results
Red light having a wavelength of 700 nm is projected onto magnesium metal to which electrons are bound by 3.68 eV. (a) Use ${\text{KE}}_{e}= hf-\text{BE}$ to calculate the kinetic energy of the ejected electrons. (b) What is unreasonable about this result? (c) Which assumptions are unreasonable or inconsistent?
(a) $-1.90 \text{eV}$ (b) Negative kinetic energy
(c) That the electrons would be knocked free.
Unreasonable Results
(a) What is the binding energy of electrons to a material from which 4.00-eV electrons are ejected by 400-nm EM radiation? (b) What is unreasonable about this result? (c) Which assumptions are unreasonable or inconsistent?
Strategy
Calculate the photon energy, then use the photoelectric equation to find the binding energy. Examine whether the result is physically reasonable.
Solution for (a)
Calculate the photon energy:
Using the photoelectric equation:
Solution for (b)
A negative binding energy is physically impossible. Binding energy represents the energy required to remove an electron from a material, which must be positive. A negative value would imply the electron is repelled by the material, contradicting the definition of binding.
Solution for (c)
The assumption that is unreasonable is that 400-nm photons can eject electrons with 4.00 eV of kinetic energy. The photon energy (3.10 eV) is less than the claimed kinetic energy of the ejected electrons (4.00 eV). By conservation of energy, this is impossible—the electron cannot have more kinetic energy than the total energy provided by the photon. Either the photon wavelength must be shorter (higher energy), or the electron kinetic energy must be less than 3.10 eV.
(a) −0.90 eV
(b) Negative binding energy is physically impossible
(c) The assumption that 400-nm photons (3.10 eV) can produce 4.00-eV electrons violates energy conservation