
Magnetic force can cause a charged particle to move in a circular or spiral path. Cosmic rays are energetic charged particles in outer space, some of which approach the Earth. They can be forced into spiral paths by the Earth’s magnetic field. Protons in giant accelerators are kept in a circular path by magnetic force. The bubble chamber photograph in [Figure 1] shows charged particles moving in such curved paths. The curved paths of charged particles in magnetic fields are the basis of a number of phenomena and can even be used analytically, such as in a mass spectrometer.

So does the magnetic force cause circular motion? Magnetic force is always perpendicular to velocity, so that it does no work on the charged particle. The particle’s kinetic energy and speed thus remain constant. The direction of motion is affected, but not the speed. This is typical of uniform circular motion. The simplest case occurs when a charged particle moves perpendicular to a uniform $B$ -field, such as shown in [Figure 2]. (If this takes place in a vacuum, the magnetic field is the dominant factor determining the motion.) Here, the magnetic force supplies the centripetal force ${F}_{c}=mv^{2}/r$ . Noting that $\sin \theta =1$ , we see that $F=qvB$.
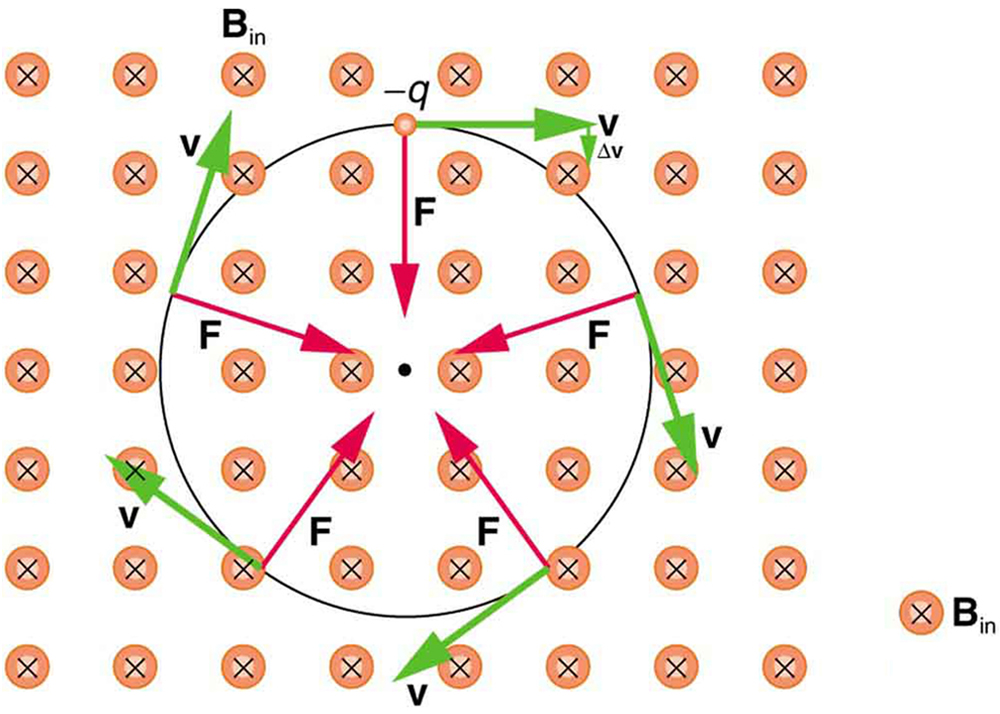
Because the magnetic force $F$ supplies the centripetal force ${F}_{c}$ , we have
Solving for $r$ yields
Here, $r$ is the radius of curvature of the path of a charged particle with mass $m$ and charge $q$ , moving at a speed $v$ perpendicular to a magnetic field of strength $B$ . If the velocity is not perpendicular to the magnetic field, then $v$ is the component of the velocity perpendicular to the field. The component of the velocity parallel to the field is unaffected, since the magnetic force is zero for motion parallel to the field. This produces a spiral motion rather than a circular one.
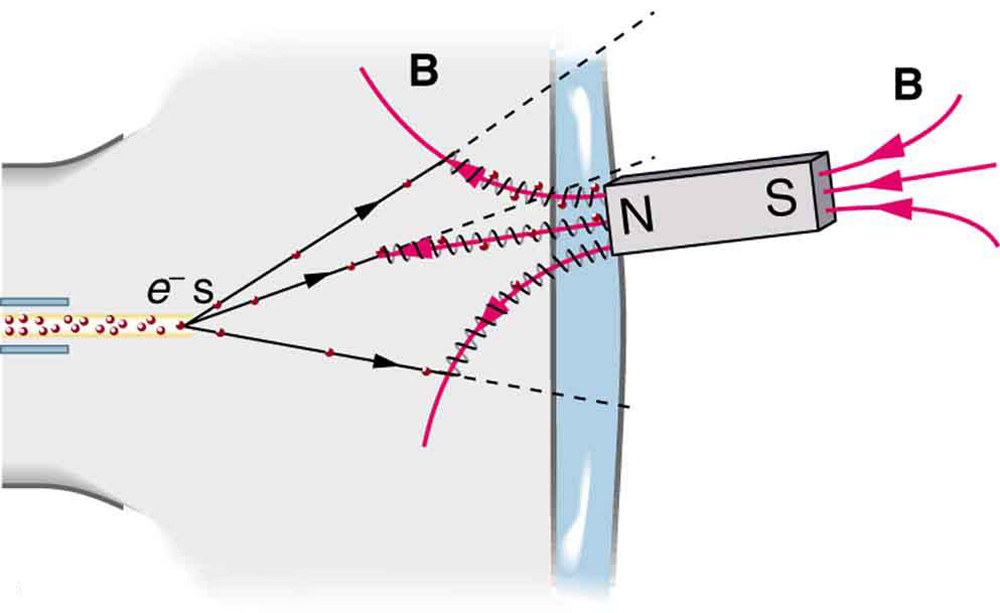
A magnet brought near an old-fashioned TV screen such as in [Figure 3] (TV sets with cathode ray tubes instead of LCD screens) severely distorts its picture by altering the path of the electrons that make its phosphors glow. **(Don’t try this at home, as it will permanently magnetize and ruin the TV.) ** To illustrate this, calculate the radius of curvature of the path of an electron having a velocity of $6.00 \times 10^{7} \text{m/s}$ (corresponding to the accelerating voltage of about 10.0 kV used in some TVs) perpendicular to a magnetic field of strength $B=0.500\text{ T}$ (obtainable with permanent magnets).
Strategy
We can find the radius of curvature $r$ directly from the equation $r=\frac{mv}{qB}$ , since all other quantities in it are given or known.
Solution
Using known values for the mass and charge of an electron, along with the given values of $v$ and $B$ gives us
or
Discussion
The small radius indicates a large effect. The electrons in the TV picture tube are made to move in very tight circles, greatly altering their paths and distorting the image.
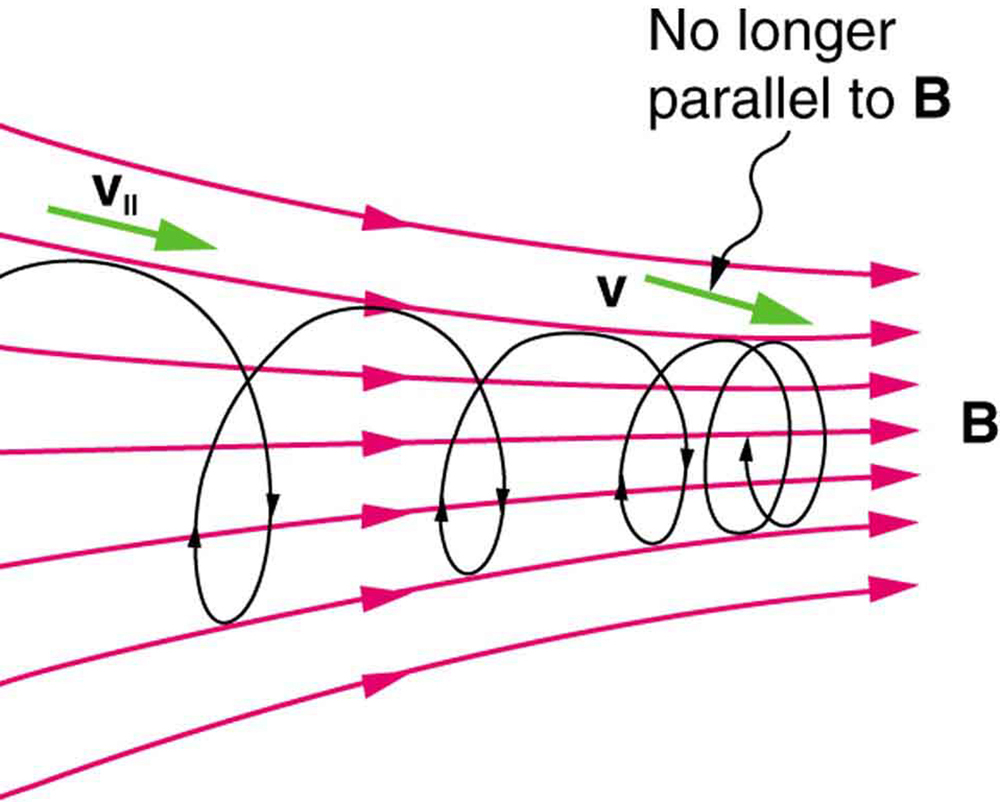
[Figure 4] shows how electrons not moving perpendicular to magnetic field lines follow the field lines. The component of velocity parallel to the lines is unaffected, and so the charges spiral along the field lines. If field strength increases in the direction of motion, the field will exert a force to slow the charges, forming a kind of magnetic mirror, as shown below.
The properties of charged particles in magnetic fields are related to such different things as the Aurora Australis or Aurora Borealis and particle accelerators. Charged particles approaching magnetic field lines may get trapped in spiral orbits about the lines rather than crossing them, as seen in [Figure 5]. Some cosmic rays, for example, follow the Earth’s magnetic field lines, entering the atmosphere near the magnetic poles and causing the southern or northern lights through their ionization of molecules in the atmosphere. This glow of energized atoms and molecules is seen in [Figure 1]. Those particles that approach middle latitudes must cross magnetic field lines, and many are prevented from penetrating the atmosphere. Cosmic rays are a component of background radiation; consequently, they give a higher radiation dose at the poles than at the equator.
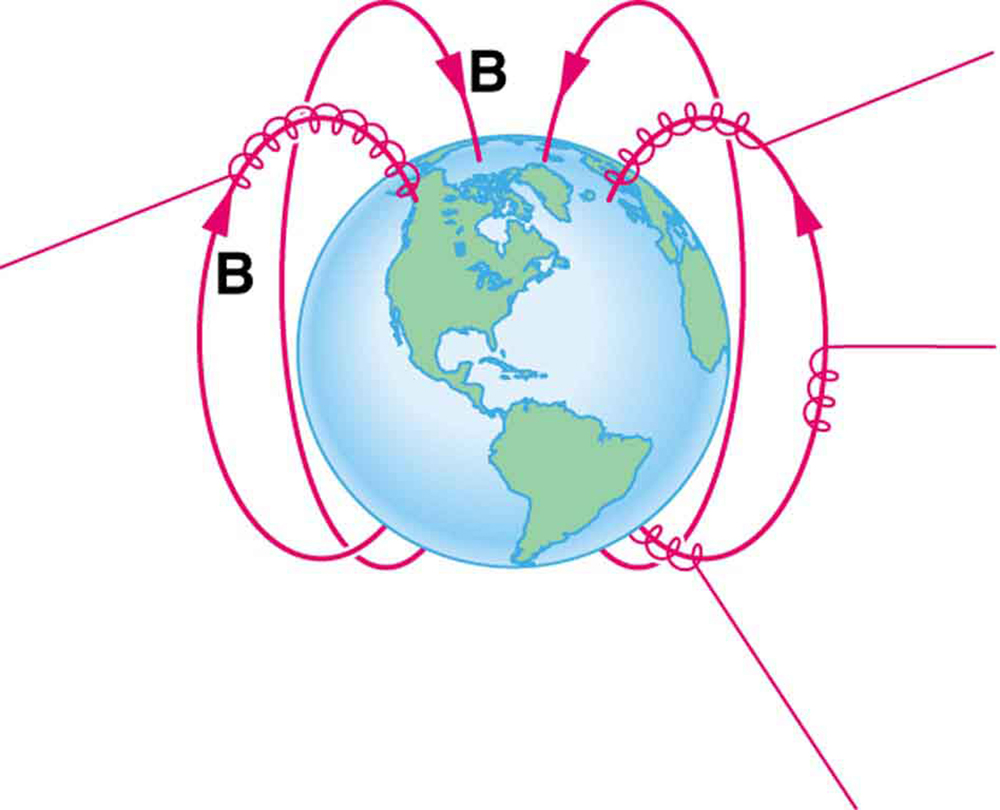
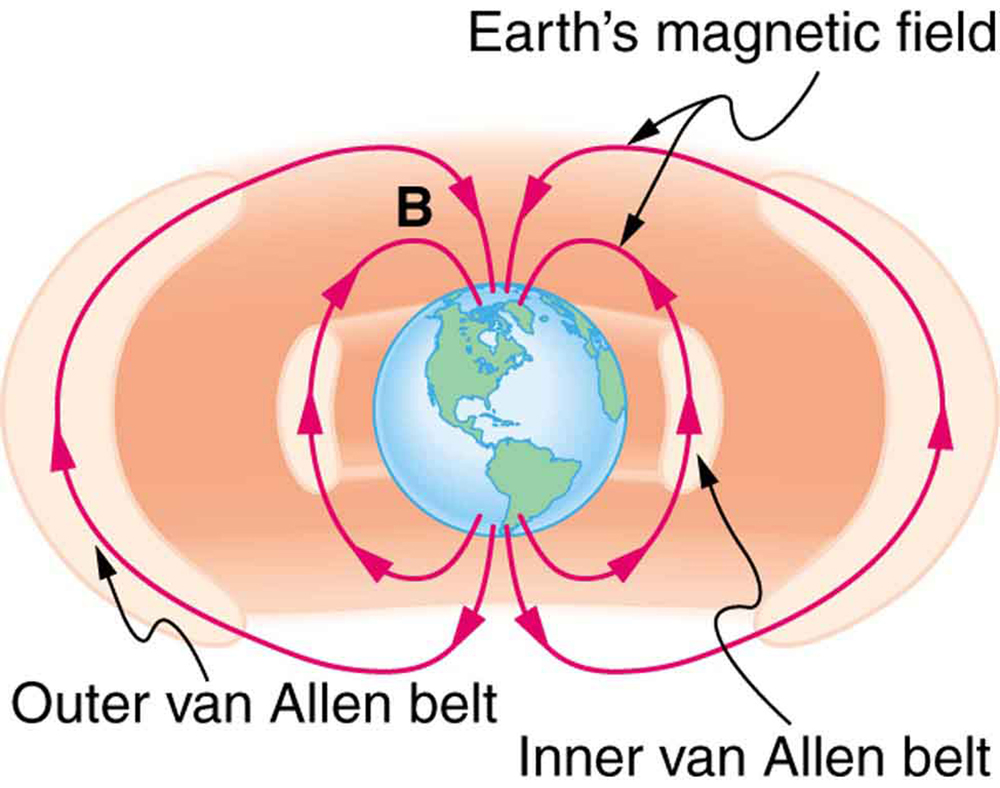
Some incoming charged particles become trapped in the Earth’s magnetic field, forming two belts above the atmosphere known as the Van Allen radiation belts after the discoverer James A. Van Allen, an American astrophysicist. ( See [Figure 6].) Particles trapped in these belts form radiation fields (similar to nuclear radiation) so intense that manned space flights avoid them and satellites with sensitive electronics are kept out of them. In the few minutes it took lunar missions to cross the Van Allen radiation belts, astronauts received radiation doses more than twice the allowed annual exposure for radiation workers. Other planets have similar belts, especially those having strong magnetic fields like Jupiter.
Back on Earth, we have devices that employ magnetic fields to contain charged particles. Among them are the giant particle accelerators that have been used to explore the substructure of matter. (See [Figure 7].) Magnetic fields not only control the direction of the charged particles, they also are used to focus particles into beams and overcome the repulsion of like charges in these beams.

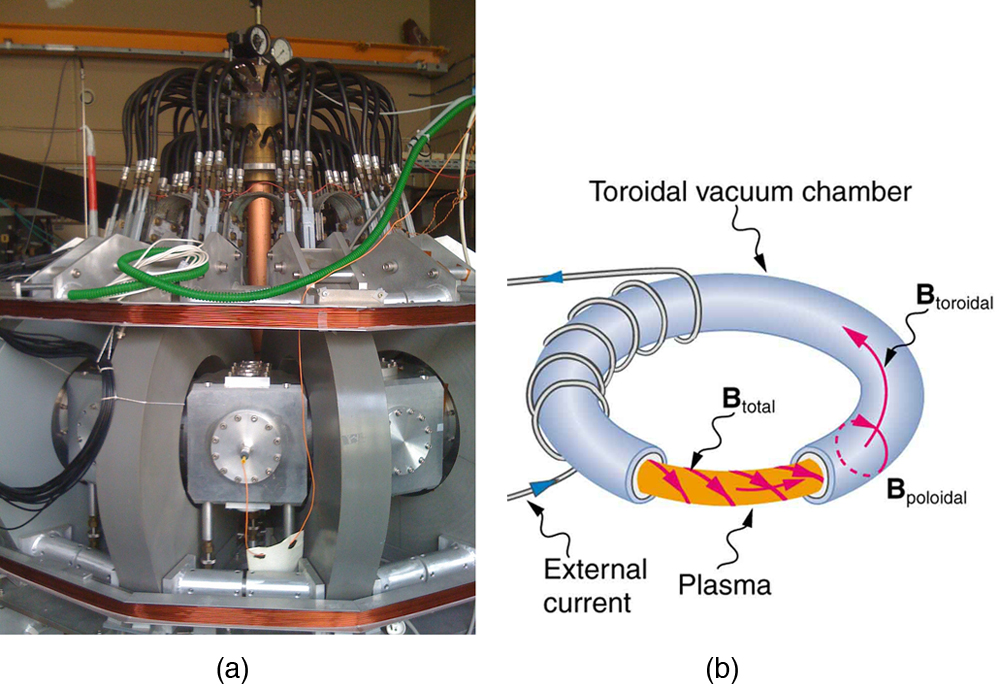
Thermonuclear fusion (like that occurring in the Sun) is a hope for a future clean energy source. One of the most promising devices is the tokamak, which uses magnetic fields to contain (or trap) and direct the reactive charged particles. (See [Figure 8].) Less exotic, but more immediately practical, amplifiers in microwave ovens use a magnetic field to contain oscillating electrons. These oscillating electrons generate the microwaves sent into the oven.
Mass spectrometers have a variety of designs, and many use magnetic fields to measure mass. The curvature of a charged particle’s path in the field is related to its mass and is measured to obtain mass information. ( See More Applications of Magnetism.) Historically, such techniques were employed in the first direct observations of electron charge and mass. Today, mass spectrometers (sometimes coupled with gas chromatographs) are used to determine the make-up and sequencing of large biological molecules.
where $v$ is the component of the velocity perpendicular to $B$ for a charged particle with mass $m$ and charge $q$.
How can the motion of a charged particle be used to distinguish between a magnetic and an electric field?
High-velocity charged particles can damage biological cells and are a component of radiation exposure in a variety of locations ranging from research facilities to natural background. Describe how you could use a magnetic field to shield yourself.
If a cosmic ray proton approaches the Earth from outer space along a line toward the center of the Earth that lies in the plane of the equator, in what direction will it be deflected by the Earth’s magnetic field? What about an electron? A neutron?
What are the signs of the charges on the particles in [Figure 9]?
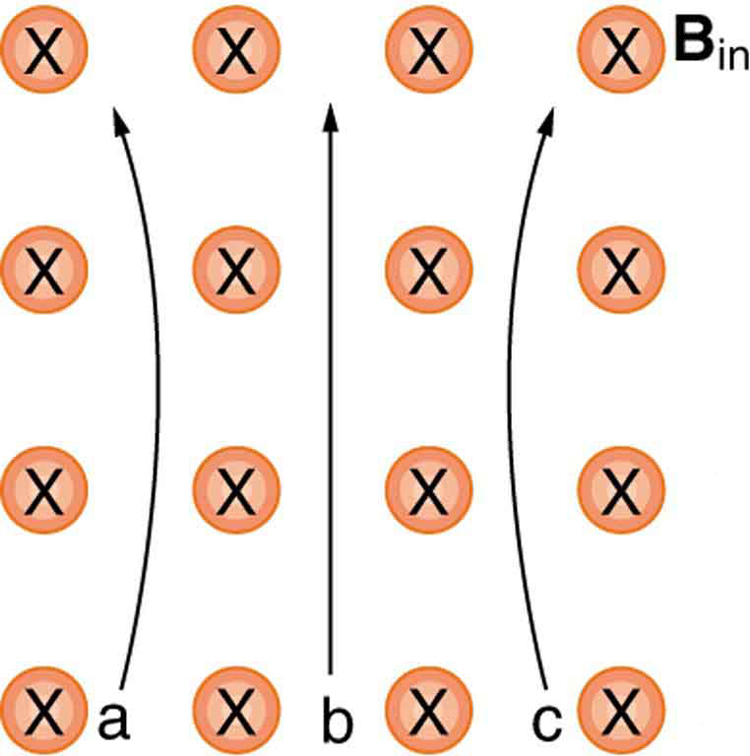
Which of the particles in [Figure 10] has the greatest velocity, assuming they have identical charges and masses?
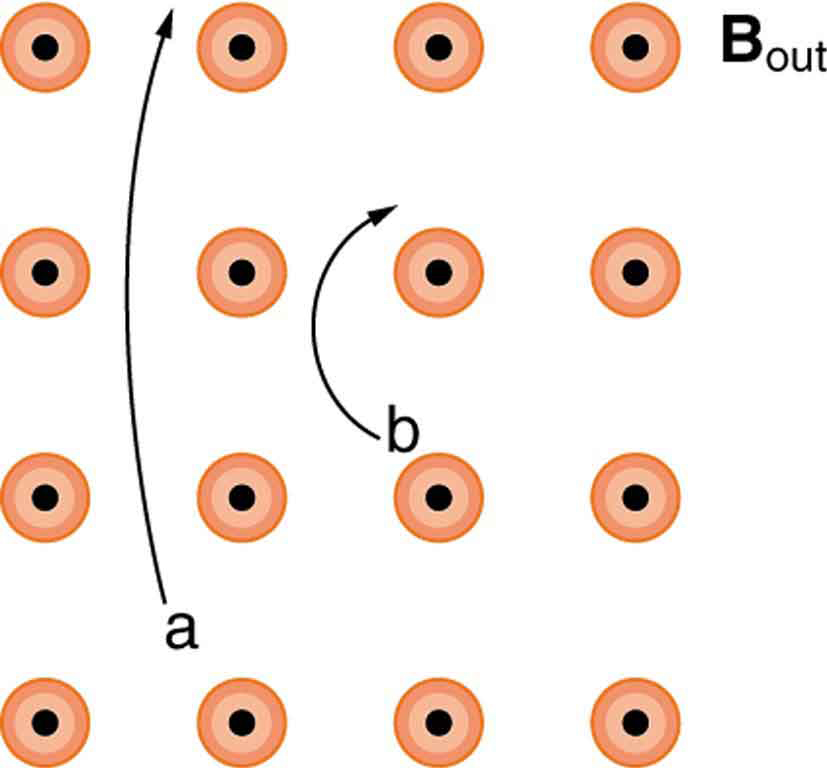
Which of the particles in [Figure 9] has the greatest mass, assuming all have identical charges and velocities?
While operating, a high-precision TV monitor is placed on its side during maintenance. The image on the monitor changes color and blurs slightly. Discuss the possible relation of these effects to the Earth’s magnetic field.
If you need additional support for these problems, see More Applications of Magnetism.
A cosmic ray electron moves at $7.50 \times 10^{6} \text{m/s}$ perpendicular to the Earth’s magnetic field at an altitude where field strength is $1.00 \times 10^{-5} T$ . What is the radius of the circular path the electron follows?
Strategy
When a charged particle moves perpendicular to a uniform magnetic field, the magnetic force provides the centripetal force for circular motion. We equate the magnetic force $F = qvB$ to the centripetal force $F_c = mv^2/r$ and solve for the radius $r = mv/(qB)$.
Solution
Known quantities:
| Electron charge: $$ | q | = e = 1.60 \times 10^{-19} \text{ C}$$ |
Unknown: Radius $r$
Using the circular motion radius formula:
Discussion
This radius of about 4.3 meters is quite large—much larger than what we’d observe in a laboratory setting with stronger magnets. This is because Earth’s magnetic field at high altitude (where the problem specifies $B = 1.00 \times 10^{-5}$ T, weaker than at the surface) is relatively weak. Cosmic ray electrons indeed follow curved paths of this scale in Earth’s magnetic field, which is part of how they become trapped in the Van Allen radiation belts. In particle physics laboratories, much stronger fields (1-10 T) are used to bend particle paths into tight curves for detection.
The radius of the electron’s circular path is 4.27 m.
A proton moves at $7.50 \times 10^{7} \text{m/s}$ perpendicular to a magnetic field. The field causes the proton to travel in a circular path of radius 0.800 m. What is the field strength?
Strategy
We use the same relationship between radius, velocity, charge, mass, and magnetic field: $r = mv/(qB)$. Here we know the radius and velocity and need to solve for the magnetic field strength $B$.
Solution
Known quantities:
Unknown: Magnetic field strength $B$
From $r = mv/(qB)$, solving for $B$:
Discussion
A field strength just under 1 T is readily achievable with modern permanent magnets or electromagnets. The proton is moving at 25% of the speed of light, which is very fast but not so fast that relativistic corrections are essential (though they would improve accuracy). This type of setup—high-speed protons in a strong magnetic field—is the basis of cyclotrons and synchrotrons used in particle physics and medical proton therapy.
The magnetic field strength is 0.977 T (approximately 0.98 T).
(a) Viewers of Star Trek hear of an antimatter drive on the Starship Enterprise. One possibility for such a futuristic energy source is to store antimatter charged particles in a vacuum chamber, circulating in a magnetic field, and then extract them as needed. Antimatter annihilates with normal matter, producing pure energy. What strength magnetic field is needed to hold antiprotons, moving at $5.00 \times 10^{7} \text{m/s}$ in a circular path 2.00 m in radius? Antiprotons have the same mass as protons but the opposite (negative) charge. (b) Is this field strength obtainable with today’s technology or is it a futuristic possibility?
Strategy
| Antiprotons have the same mass as protons but opposite (negative) charge. The magnitude of the charge is still $e$. We use $$r = mv/( | q | B)$and solve for$B$$. For part (b), we compare the required field strength to what can be achieved with current technology. |
Solution
Known quantities:
| Antiproton charge magnitude: $$ | q | = e = 1.60 \times 10^{-19} \text{ C}$$ |
(a) Required magnetic field:
(b) Technological feasibility:
A field of 0.261 T is easily achievable with today’s technology:
This is well within the capabilities of even relatively inexpensive magnets.
Discussion
Antimatter storage is actually done at facilities like CERN, where antiprotons and positrons are stored in magnetic “bottles” called Penning traps and storage rings. The challenge isn’t the magnetic field strength but rather maintaining the ultra-high vacuum needed to prevent annihilation with residual gas molecules, and the difficulty of producing significant quantities of antimatter in the first place. The curvature direction for antiprotons would be opposite to protons in the same field because of their negative charge.
(a) The required magnetic field strength is 0.261 T.
(b) This field strength is readily obtainable with current technology. Even permanent magnets can produce fields of 0.5 T or more, so containing antiprotons magnetically is technologically feasible today.
(a) An oxygen-16 ion with a mass of $2.66 \times 10^{-26} \text{kg}$ travels at $5.00 \times 10^{6} \text{m/s}$ perpendicular to a 1.20-T magnetic field, which makes it move in a circular arc with a 0.231-m radius. What positive charge is on the ion? (b) What is the ratio of this charge to the charge of an electron? (c) Discuss why the ratio found in (b) should be an integer.
Strategy
We use the circular motion equation $r = mv/(qB)$ and solve for the charge $q$. Then we compare this charge to the elementary charge to find the ionization state.
Solution
Known quantities:
(a) Charge on the ion:
From $r = mv/(qB)$, solving for $q$:
(b) Ratio to electron charge:
(c) Why the ratio should be an integer:
Electric charge is quantized—it comes only in integer multiples of the elementary charge $e$. This is because charge carriers are discrete particles:
The oxygen-16 ion has lost 3 electrons, making it O$^{3+}$ (triply ionized oxygen). Fractional charges are not observed for isolated particles in nature. The slight deviation from exactly 3.00 in calculations is due to rounding in the given values.
Discussion
Mass spectrometers exploit this quantization of charge. Since $r \propto m/q$, ions with different charge states will follow different radii, allowing them to be separated. The O$^{3+}$ ion would follow a different path than O$^{2+}$ or O$^{+}$ in the same field, even though they have the same mass.
(a) The charge on the ion is $4.80 \times 10^{-19}$ C.
(b) The ratio of this charge to the electron charge is 3.00 (the ion is triply ionized: O$^{3+}$).
(c) The ratio must be an integer because electric charge is quantized—charges exist only in integer multiples of the elementary charge $e$, corresponding to the number of electrons removed from or added to an atom.
What radius circular path does an electron travel if it moves at the same speed and in the same magnetic field as the proton in [Exercise2]?
Strategy
The radius of circular motion is $r = mv/(qB)$. Since the electron has the same charge magnitude as the proton but much smaller mass (about 1/1836 of the proton mass), it will travel in a much smaller circle at the same speed and in the same field.
Solution
Known quantities (from Exercise 2):
| Electron charge magnitude: $$ | q | = e = 1.60 \times 10^{-19} \text{ C}$$ |
Discussion
The electron’s radius is about 1836 times smaller than the proton’s 0.800 m radius. This ratio equals the proton-to-electron mass ratio, which makes sense since $r \propto m$ when $v$, $q$, and $B$ are the same. This dramatic difference in path radius is exploited in mass spectrometers to separate particles by mass. The electron’s tiny radius (less than half a millimeter) would require very precise measurement to detect the curvature.
The electron would travel in a circular path with radius $4.36 \times 10^{-4}$ m (0.436 mm).
A velocity selector in a mass spectrometer uses a 0.100-T magnetic field. (a) What electric field strength is needed to select a speed of $4.00 \times 10^{6} \text{m/s}$ ? (b) What is the voltage between the plates if they are separated by 1.00 cm?
Strategy
A velocity selector uses crossed electric and magnetic fields. Charged particles experience both forces, but only those with a specific velocity travel straight through—when the electric force equals the magnetic force: $qE = qvB$. This gives the selection condition $v = E/B$. The voltage is then found from $V = Ed$ for a uniform field between parallel plates.
Solution
Known quantities:
(a) Electric field strength:
From the velocity selector condition $v = E/B$:
(b) Voltage between plates:
Discussion
The velocity selector is a crucial component of mass spectrometers because it ensures all ions entering the magnetic separation region have the same speed. Without this, particles with different speeds but the same mass-to-charge ratio would follow different paths, reducing the resolution of the instrument. A voltage of 4 kV across a 1 cm gap is substantial but manageable in laboratory settings. Note that the selection condition is independent of the charge and mass of the particles—only their velocity determines whether they pass through undeflected.
(a) The electric field strength needed is $4.00 \times 10^{5}$ V/m (400 kV/m).
(b) The voltage between the plates is 4.00 kV (4000 V).
An electron in a TV CRT moves with a speed of $6.00 \times 10^{7} \text{m/s}$ , in a direction perpendicular to the Earth’s field, which has a strength of $5.00 \times 10^{-5} \text{T}$ . (a) What strength electric field must be applied perpendicular to the Earth’s field to make the electron moves in a straight line? (b) If this is done between plates separated by 1.00 cm, what is the voltage applied? (Note that TVs are usually surrounded by a ferromagnetic material to shield against external magnetic fields and avoid the need for such a correction.)
Strategy
This is similar to the velocity selector problem. To make the electron travel in a straight line, the electric force must balance the magnetic force: $eE = evB$, giving $E = vB$. The voltage is then $V = Ed$.
Solution
Known quantities:
(a) Electric field strength:
For the electron to travel in a straight line, the electric force must equal the magnetic force:
(b) Voltage between plates:
Discussion
This relatively modest voltage of 30 V (less than that of a typical laptop charger) would be needed to counteract Earth’s magnetic field effect on the electron beam. However, TVs were typically surrounded by ferromagnetic shielding (mu-metal) to reduce the influence of external magnetic fields, making this correction unnecessary in practice. The shielding approach is more practical than actively applying correcting fields, especially since Earth’s field direction varies with the TV’s orientation.
(a) The electric field strength needed is 3.00 kV/m (3000 V/m).
(b) The voltage between the plates is 30.0 V.
(a) At what speed will a proton move in a circular path of the same radius as the electron in [Exercise1]? (b) What would the radius of the path be if the proton had the same speed as the electron? (c) What would the radius be if the proton had the same kinetic energy as the electron? (d) The same momentum?
Strategy
We use $r = mv/(qB)$ for all parts. For particles with the same charge magnitude, the radius depends on the mass-velocity product (momentum). We’ll compare the proton and electron under different conditions specified in each part.
Solution
Reference values from Exercise 1:
(a) Proton speed for same radius:
For the same radius with $r = mv/(qB)$ and same $q$ and $B$:
For $r_p = r_e$:
(b) Proton radius at same speed:
(c) Proton radius at same kinetic energy:
If $\frac{1}{2}m_p v_p^2 = \frac{1}{2}m_e v_e^2$, then $v_p = v_e\sqrt{m_e/m_p}$
(d) Proton radius at same momentum:
If $m_p v_p = m_e v_e$, then $v_p = v_e(m_e/m_p)$
Discussion
These results reveal important physics:
(a) The proton speed would be $4.08 \times 10^{3}$ m/s (about 4 km/s).
(b) The proton radius would be 7.84 km (7840 m).
(c) The proton radius with equal kinetic energy would be 183 m.
(d) The proton radius with equal momentum would be 4.27 m (same as the electron).
A mass spectrometer is being used to separate common oxygen-16 from the much rarer oxygen-18, taken from a sample of old glacial ice. (The relative abundance of these oxygen isotopes is related to climatic temperature at the time the ice was deposited.) The ratio of the masses of these two ions is 16 to 18, the mass of oxygen-16 is $2.66 \times 10^{-26} \text{kg},$ and they are singly charged and travel at $5.00 \times 10^{6} \text{m/s}$ in a 1.20-T magnetic field. What is the separation between their paths when they hit a target after traversing a semicircle?
Strategy
In a mass spectrometer, ions with the same charge and velocity but different masses follow circular paths with different radii. Since $r = mv/(qB)$, heavier ions follow larger-radius paths. After a semicircle, ions are separated by the difference in their diameters: separation = $2(r_{18} - r_{16})$.
Solution
Known quantities:
Calculate the radii:
Separation after semicircle:
Discussion
The separation of 17.3 cm is quite large and easily measurable. This is why mass spectrometry can distinguish isotopes with high precision. The key factor is the mass difference (12.5% between O-16 and O-18), which produces a proportional difference in radius. Paleoclimatologists use oxygen isotope ratios in ice cores to reconstruct ancient temperatures because the ratio of O-18 to O-16 in precipitation varies with temperature.
The separation between the oxygen-16 and oxygen-18 paths is 0.173 m (17.3 cm).
(a) Triply charged uranium-235 and uranium-238 ions are being separated in a mass spectrometer. (The much rarer uranium-235 is used as reactor fuel.) The masses of the ions are $3.90 \times 10^{-25} \text{kg}$ and $3.95 \times 10^{-25} \text{kg}$ , respectively, and they travel at $3.00 \times 10^{5} \text{m/s}$ in a 0.250-T field. What is the separation between their paths when they hit a target after traversing a semicircle? (b) Discuss whether this distance between their paths seems to be big enough to be practical in the separation of uranium-235 from uranium-238.
Strategy
This is similar to the oxygen isotope problem, but with triply charged ions ($q = 3e$) and heavier masses. We calculate the radius for each isotope using $r = mv/(qB)$, then find the separation as the difference in diameters.
Solution
Known quantities:
(a) Calculate the radii:
Separation after semicircle:
(b) Practical considerations:
A separation of 2.5 cm is large enough to be practical for separating uranium isotopes. This is detectable and sufficient for directing the ion beams into separate collectors. However, several challenges make large-scale separation difficult:
During World War II, electromagnetic separation (calutrons) using this principle was one method used to enrich uranium for the first atomic bombs at Oak Ridge, Tennessee. Today, gas centrifuge technology is more efficient for large-scale enrichment.
Discussion
The small mass difference between U-235 and U-238 (compared to oxygen isotopes) results in smaller separation. However, 2.5 cm is still workable with precision engineering. Modern mass spectrometers can resolve mass differences much smaller than this.
(a) The separation between U-235 and U-238 paths is 0.025 m (2.5 cm).
(b) Yes, 2.5 cm is large enough to be practical for isotope separation. While challenging, electromagnetic separation was historically used to enrich uranium, and this separation distance is sufficient for collecting the isotopes separately.