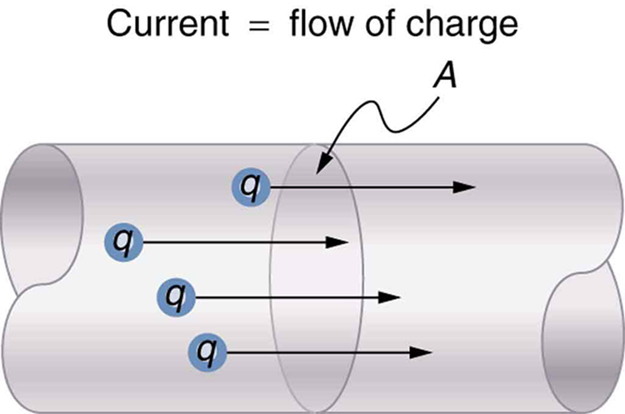
Electric current is defined to be the rate at which charge flows. A large current, such as that used to start a truck engine, moves a large amount of charge in a small time, whereas a small current, such as that used to operate a hand-held calculator, moves a small amount of charge over a long period of time. In equation form, electric current $I$ is defined to be
where $\Delta Q$ is the amount of charge passing through a given area in time $\Delta t$ . (As in previous chapters, initial time is often taken to be zero, in which case $\Delta t=t$ .) (See [Figure 1].) The SI unit for current is the ampere (A), named for the French physicist André-Marie Ampère (1775–1836). Since $I=\Delta Q/\Delta t$ , we see that an ampere is one coulomb per second:
Not only are fuses and circuit breakers rated in amperes (or amps), so are many electrical appliances.

(a) What is the current involved when a truck battery sets in motion 720 C of charge in 4.00 s while starting an engine? (b) How long does it take 1.00 C of charge to flow through a handheld calculator if a 0.300-mA current is flowing?
Strategy
We can use the definition of current in the equation $I=\Delta Q/\Delta t$ to find the current in part (a), since charge and time are given. In part (b), we rearrange the definition of current and use the given values of charge and current to find the time required.
Solution for (a)
Entering the given values for charge and time into the definition of current gives
Discussion for (a)
This large value for current illustrates the fact that a large charge is moved in a small amount of time. The currents in these “starter motors” are fairly large because large frictional forces need to be overcome when setting something in motion.
Solution for (b)
Solving the relationship $I=\Delta Q/\Delta t$ for time $\Delta t$ , and entering the known values for charge and current gives
Discussion for (b)
This time is slightly less than an hour. The small current used by the hand-held calculator takes a much longer time to move a smaller charge than the large current of the truck starter. So why can we operate our calculators only seconds after turning them on? It’s because calculators require very little energy. Such small current and energy demands allow handheld calculators to operate from solar cells or to get many hours of use out of small batteries. Remember, calculators do not have moving parts in the same way that a truck engine has with cylinders and pistons, so the technology requires smaller currents.
[Figure 2] shows a simple circuit and the standard schematic representation of a battery, conducting path, and load (a resistor). Schematics are very useful in visualizing the main features of a circuit. A single schematic can represent a wide variety of situations. The schematic in [Figure 2] (b), for example, can represent anything from a truck battery connected to a headlight lighting the street in front of the truck to a small battery connected to a penlight lighting a keyhole in a door. Such schematics are useful because the analysis is the same for a wide variety of situations. We need to understand a few schematics to apply the concepts and analysis to many more situations.
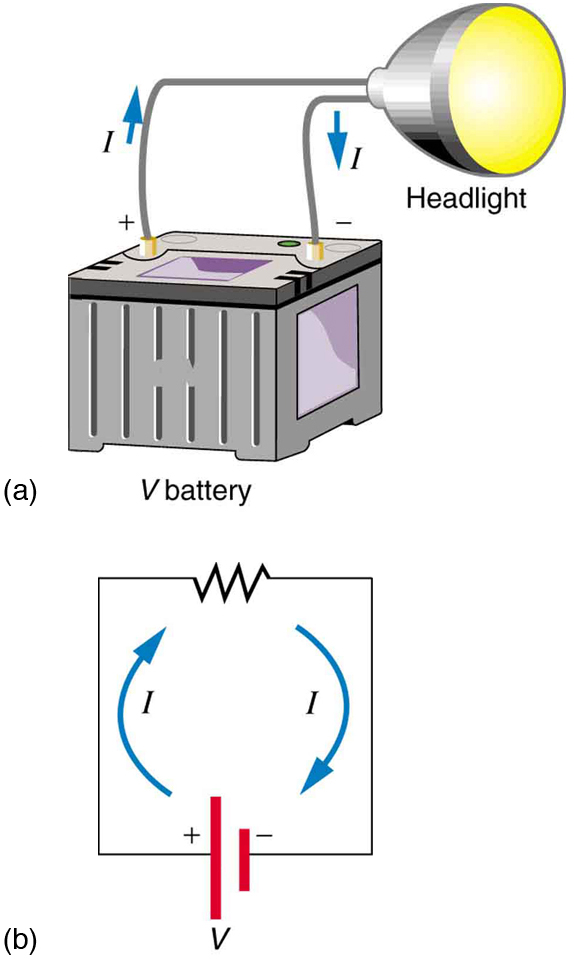
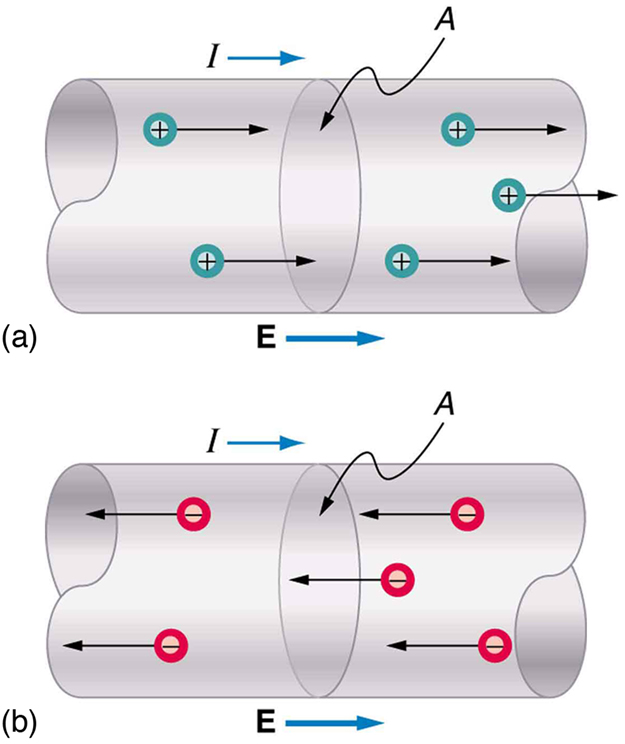
Note that the direction of current flow in [Figure 2] is from positive to negative. The direction of conventional current is the direction that positive charge would flow. Depending on the situation, positive charges, negative charges, or both may move. In metal wires, for example, current is carried by electrons—that is, negative charges move. In ionic solutions, such as salt water, both positive and negative charges move. This is also true in nerve cells. A Van de Graaff generator used for nuclear research can produce a current of pure positive charges, such as protons. [Figure 3] illustrates the movement of charged particles that compose a current. The fact that conventional current is taken to be in the direction that positive charge would flow can be traced back to American politician and scientist Benjamin Franklin in the 1700s. He named the type of charge associated with electrons negative, long before they were known to carry current in so many situations. Franklin, in fact, was totally unaware of the small-scale structure of electricity.
It is important to realize that there is an electric field in conductors responsible for producing the current, as illustrated in [Figure 3]. Unlike static electricity, where a conductor in equilibrium cannot have an electric field in it, conductors carrying a current have an electric field and are not in static equilibrium. An electric field is needed to supply energy to move the charges.
Find a straw and little peas that can move freely in the straw. Place the straw flat on a table and fill the straw with peas. When you pop one pea in at one end, a different pea should pop out the other end. This demonstration is an analogy for an electric current. Identify what compares to the electrons and what compares to the supply of energy. What other analogies can you find for an electric current?
Note that the flow of peas is based on the peas physically bumping into each other; electrons flow due to mutually repulsive electrostatic forces.
If the 0.300-mA current through the calculator mentioned in the [Example 1] example is carried by electrons, how many electrons per second pass through it?
Strategy
The current calculated in the previous example was defined for the flow of positive charge. For electrons, the magnitude is the same, but the sign is opposite, ${I}_{\text{electrons}}=-0.300 \times 10^{-3 }\text{C/s}$. Since each electron $\left({e}^{-}\right)$ has a charge of $- 1.60 \times 10^{-19} \text{C}$ , we can convert the current in coulombs per second to electrons per second.
Solution
Starting with the definition of current, we have
We divide this by the charge per electron, so that
Discussion
There are so many charged particles moving, even in small currents, that individual charges are not noticed, just as individual water molecules are not noticed in water flow. Even more amazing is that they do not always keep moving forward like soldiers in a parade. Rather they are like a crowd of people with movement in different directions but a general trend to move forward. There are lots of collisions with atoms in the metal wire and, of course, with other electrons.
Electrical signals are known to move very rapidly. Telephone conversations carried by currents in wires cover large distances without noticeable delays. Lights come on as soon as a switch is flicked. Most electrical signals carried by currents travel at speeds on the order of $10^{8} \text{m/s}$ , a significant fraction of the speed of light. Interestingly, the individual charges that make up the current move much more slowly on average, typically drifting at speeds on the order of $10^{-4} \text{m/s}$ . How do we reconcile these two speeds, and what does it tell us about standard conductors?
The high speed of electrical signals results from the fact that the force between charges acts rapidly at a distance. Thus, when a free charge is forced into a wire, as in [Figure 4], the incoming charge pushes other charges ahead of it, which in turn push on charges farther down the line. The density of charge in a system cannot easily be increased, and so the signal is passed on rapidly. The resulting electrical shock wave moves through the system at nearly the speed of light. To be precise, this rapidly moving signal or shock wave is a rapidly propagating change in electric field.
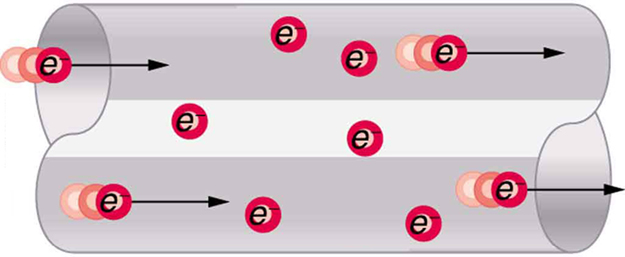
Good conductors have large numbers of free charges in them. In metals, the free charges are free electrons. [Figure 5] shows how free electrons move through an ordinary conductor. The distance that an individual electron can move between collisions with atoms or other electrons is quite small. The electron paths thus appear nearly random, like the motion of atoms in a gas. But there is an electric field in the conductor that causes the electrons to drift in the direction shown (opposite to the field, since they are negative). The drift velocity ${v}_{\text{d}}$ is the average velocity of the free charges. Drift velocity is quite small, since there are so many free charges. If we have an estimate of the density of free electrons in a conductor, we can calculate the drift velocity for a given current. The larger the density, the lower the velocity required for a given current.
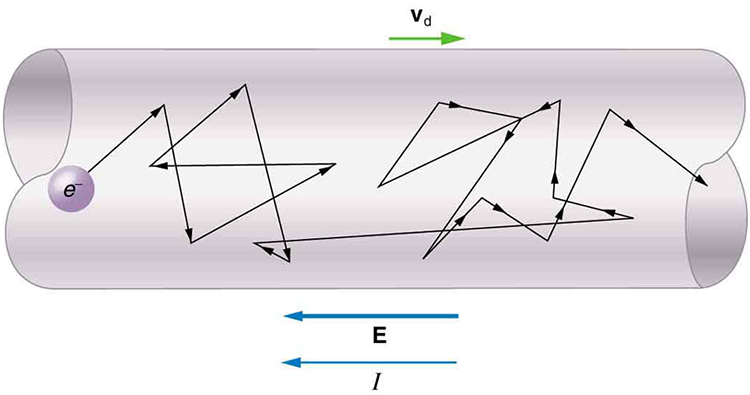
Good electrical conductors are often good heat conductors, too. This is because large numbers of free electrons can carry electrical current and can transport thermal energy.
The free-electron collisions transfer energy to the atoms of the conductor. The electric field does work in moving the electrons through a distance, but that work does not increase the kinetic energy (nor speed, therefore) of the electrons. The work is transferred to the conductor’s atoms, possibly increasing temperature. Thus a continuous power input is required to keep a current flowing. An exception, of course, is found in superconductors, for reasons we shall explore in a later chapter. Superconductors can have a steady current without a continual supply of energy—a great energy savings. In contrast, the supply of energy can be useful, such as in a lightbulb filament. The supply of energy is necessary to increase the temperature of the tungsten filament, so that the filament glows.
Find a lightbulb with a filament. Look carefully at the filament and describe its structure. To what points is the filament connected?
We can obtain an expression for the relationship between current and drift velocity by considering the number of free charges in a segment of wire, as illustrated in [Figure 6]. The number of free charges per unit volume is given the symbol $n$ and depends on the material. The shaded segment has a volume $A x$ , so that the number of free charges in it is $nAx$ . The charge $\Delta Q$ in this segment is thus $qnAx$ , where $q$ is the amount of charge on each carrier. (Recall that for electrons, $q$ is $-1.60 \times 10^{-19} \text{C}$ .) Current is charge moved per unit time; thus, if all the original charges move out of this segment in time $\Delta t$ , the current is
Note that $x/\Delta t$ is the magnitude of the drift velocity, ${v}_{\text{d}}$ , since the charges move an average distance $x$ in a time $\Delta t$. Rearranging terms gives
where $I$ is the current through a wire of cross-sectional area $A$ made of a material with a free charge density $n$ . The carriers of the current each have charge $q$ and move with a drift velocity of magnitude ${v}_{\text{d}}$.
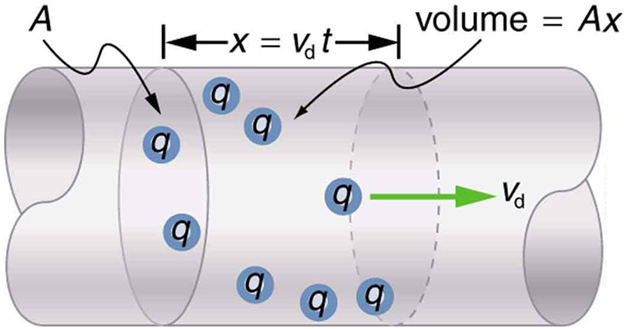
Note that simple drift velocity is not the entire story. The speed of an electron is much greater than its drift velocity. In addition, not all of the electrons in a conductor can move freely, and those that do might move somewhat faster or slower than the drift velocity. So what do we mean by free electrons? Atoms in a metallic conductor are packed in the form of a lattice structure. Some electrons are far enough away from the atomic nuclei that they do not experience the attraction of the nuclei as much as the inner electrons do. These are the free electrons. They are not bound to a single atom but can instead move freely among the atoms in a “sea” of electrons. These free electrons respond by accelerating when an electric field is applied. Of course as they move they collide with the atoms in the lattice and other electrons, generating thermal energy, and the conductor gets warmer. In an insulator, the organization of the atoms and the structure do not allow for such free electrons.
Calculate the drift velocity of electrons in a 12-gauge copper wire (which has a diameter of 2.053 mm) carrying a 20.0-A current, given that there is one free electron per copper atom. (Household wiring often contains 12-gauge copper wire, and the maximum current allowed in such wire is usually 20 A.) The density of copper is $8.80 \times 10^{3} {\text{kg/m}}^{3}$.
Strategy
We can calculate the drift velocity using the equation $I= n q A v_d$ . The current $I=20.0 \text{A}$ is given, and $q= -1.60 \times 10^{-19} \text{C}$ is the charge of an electron. We can calculate the area of a cross-section of the wire using the formula $A=\pi {r}^{2},$ where $r$ is one-half the given diameter, 2.053 mm. We are given the density of copper, $8.80 \times 10^{3} {\text{kg/m}}^{3},$ and the periodic table shows that the atomic mass of copper is 63.54 g/mol. We can use these two quantities along with Avogadro’s number, $6.02 \times 10^{23} \text{atoms/mol},$ to determine $n,$ the number of free electrons per cubic meter.
Solution
First, calculate the density of free electrons in copper. There is one free electron per copper atom. Therefore, is the same as the number of copper atoms per $\text{m}^{3}$ . We can now find $n$ as follows:
The cross-sectional area of the wire is
Rearranging $I=nqA{v}_{\text{d}}$ to isolate drift velocity gives
Discussion
The minus sign indicates that the negative charges are moving in the direction opposite to conventional current. The small value for drift velocity (on the order of $10^{-4} \text{m/s}$ ) confirms that the signal moves on the order of $10^{12}$ times faster (about $10^{8} \text{m/s}$ ) than the charges that carry it.
Electric current $I$ is the rate at which charge flows, given by
where $\Delta Q$ is the amount of charge passing through an area in time $\Delta t$ .
Current $I$ is proportional to drift velocity ${v}_{\text{d}}$ , as expressed in the relationship $I=nqAv_{\text{d}}$ . Here, $I$ is the current through a wire of cross-sectional area $A$ . The wire’s material has a free-charge density $n$ , and each carrier has charge $q$ and a drift velocity ${v}_{\text{d}}$ .
Can a wire carry a current and still be neutral—that is, have a total charge of zero? Explain.
Car batteries are rated in ampere-hours ( $\text{A}\cdot \text{h}$ ). To what physical quantity do ampere-hours correspond (voltage, charge, …), and what relationship do ampere-hours have to energy content?
If two different wires having identical cross-sectional areas carry the same current, will the drift velocity be higher or lower in the better conductor? Explain in terms of the equation ${v}_{\text{d}}=\frac{I}{nqA}$ , by considering how the density of charge carriers $n$ relates to whether or not a material is a good conductor.
Why are two conducting paths from a voltage source to an electrical device needed to operate the device?
In cars, one battery terminal is connected to the metal body. How does this allow a single wire to supply current to electrical devices rather than two wires?
Why isn’t a bird sitting on a high-voltage power line electrocuted? Contrast this with the situation in which a large bird hits two wires simultaneously with its wings.
What is the current in milliamperes produced by the solar cells of a pocket calculator through which 4.00 C of charge passes in 4.00 h?
Strategy
We use the definition of current $I = \Delta Q / \Delta t$ to find the current. We need to convert time from hours to seconds for proper SI units.
Solution
First, convert time to seconds:
Now apply the definition of current:
Discussion
This small current is typical of low-power electronic devices like calculators. The solar cells provide just enough current to operate the calculator’s circuits, which require very little power. This demonstrates why calculators can run for extended periods on minimal energy input.
The current produced by the solar cells is 0.278 mA.
A total of 600 C of charge passes through a flashlight in 0.500 h. What is the average current?
Strategy
We use the definition of electric current as the rate of charge flow: $I = \frac{\Delta Q}{\Delta t}$. The charge is given directly, but the time must be converted from hours to seconds to obtain the current in amperes.
Solution
First, convert the time to seconds:
Now apply the definition of current:
Discussion
This current of about one-third of an ampere is typical for a flashlight. It is much larger than the milliamp currents in small electronics like calculators, but much smaller than the hundreds of amperes used to start a car engine. The flashlight bulb requires enough current to heat the filament to incandescence, which demands more power than a calculator’s LED display but far less than overcoming the mechanical resistance of an engine.
The average current of 0.333 A through the flashlight is reasonable for illuminating a bulb.
What is the current when a typical static charge of $0.250 \mu \text{C}$ moves from your finger to a metal doorknob in $1.00 \mu \text{s}$ ?
Strategy
We use $I = \Delta Q / \Delta t$ with the given charge and time values. Both quantities need to be converted to SI base units.
Solution
Convert the charge and time to SI units:
Calculate the current:
Discussion
Despite the tiny amount of charge involved, the extremely short discharge time results in a significant current of 0.250 A—comparable to the current through a small lightbulb. This explains why static shocks can be felt: the momentary current is substantial enough to stimulate nerve endings. The brief duration prevents any harmful effects.
The current during the static discharge is 0.250 A.
Find the current when 2.00 nC jumps between your comb and hair over a $0.500 \text{-} \mu \text{s}$ time interval.
Strategy
This problem involves the same fundamental relationship as static discharge: $I = \frac{\Delta Q}{\Delta t}$. The charge is given in nanocoulombs and the time in microseconds, so we must convert both to SI base units (coulombs and seconds) before calculating.
Solution
Convert the given values to SI units:
Now calculate the current:
Discussion
This milliamp-level current during static discharge is consistent with the everyday experience of static shocks. Although the charge transferred is very small (nanocoulombs), the extremely short discharge time (microseconds) creates a measurable current. This brief but noticeable current is why static discharge can feel like a small shock, even though the total energy transferred is quite small.
The current produced when 2.00 nC jumps between comb and hair in 0.500 μs is 4.00 mA.
A large lightning bolt had a 20 000-A current and moved 30.0 C of charge. What was its duration?
Strategy
Rearrange the definition of current $I = \Delta Q / \Delta t$ to solve for time: $\Delta t = \Delta Q / I$.
Solution
Rearranging $I = \Delta Q / \Delta t$ for time:
Discussion
Lightning bolts transfer enormous amounts of charge (30 C here compared to microcoulombs in static shocks) in extremely short times. The 1.50 ms duration seems brief, but this is actually relatively long for lightning—the massive current and charge transfer can cause significant damage, vaporizing materials and starting fires. The energy involved (power times time) is substantial despite the short duration because the current is so high.
The lightning bolt lasted 1.50 ms.
The 200-A current through a spark plug moves 0.300 mC of charge. How long does the spark last?
Strategy
We need to find the time duration given the current and charge. Starting from the definition $I = \frac{\Delta Q}{\Delta t}$, we can solve for time: $\Delta t = \frac{\Delta Q}{I}$. The charge must be converted from milliCoulombs to Coulombs.
Solution
Convert the charge to SI units:
Solve for the time duration:
Discussion
A spark duration of 1.50 microseconds is extremely brief but physically reasonable for a spark plug. In automotive engines, spark plugs must fire rapidly and repeatedly—at high RPM, a 4-cylinder engine might require sparks at rates exceeding 100 per second per cylinder. The short duration allows the spark to initiate combustion without excessive electrode wear. The high current (200 A) during this brief interval provides enough energy to ionize the air-fuel mixture and trigger combustion, yet the total charge transferred remains small.
The spark lasts for 1.50 μs, which is sufficient to ignite the fuel-air mixture in the engine cylinder.
(a) A defibrillator sends a 6.00-A current through the chest of a patient by applying a 10 000-V potential as in the figure below. What is the resistance of the path? (b) The defibrillator paddles make contact with the patient through a conducting gel that greatly reduces the path resistance. Discuss the difficulties that would ensue if a larger voltage were used to produce the same current through the patient, but with the path having perhaps 50 times the resistance. (Hint: The current must be about the same, so a higher voltage would imply greater power. Use this equation for power: $P={I}^{2}R$ .)
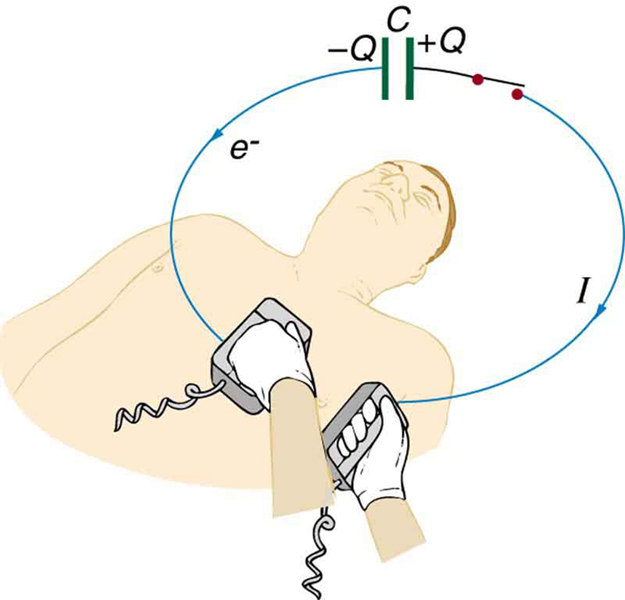
Strategy
For part (a), we use Ohm’s law $V = IR$ to find the resistance, given voltage and current. For part (b), we analyze the power dissipated using $P = I^2R$ to understand why high resistance paths are problematic.
Solution
(a) Using Ohm’s law $V = IR$, solve for resistance:
(b) With 50 times the resistance ($R' = 50R = 83.5 \text{ kΩ}$) and the same current, the voltage would need to be:
The power dissipated is:
If $R$ increases by a factor of 50 while $I$ remains constant, the power increases by a factor of 50:
Original power:
New power:
Discussion
Part (a): The resistance of 1.67 kΩ represents the path through the patient’s chest, including skin, muscle, bone, and other tissues. The conducting gel used with defibrillator paddles is essential for achieving this relatively low resistance.
Part (b): Without conducting gel, skin resistance alone can exceed 100 kΩ. If the resistance were 50 times higher (~83.5 kΩ), several severe problems would occur:
Extreme voltage required: 501 kV would be needed—far beyond safe or practical limits. This voltage would cause arcing, pose electrocution hazards to medical personnel, and likely kill the patient through excessive current or burns.
Dangerous power dissipation: The power of 3.0 MW (even if only for milliseconds) would cause catastrophic burns. Most of this energy would be dissipated in the high-resistance skin and superficial tissues, not reaching the heart.
Ineffective defibrillation: The current path would be poorly defined, with most current taking surface paths rather than passing through the heart muscle.
The conducting gel reduces skin resistance from ~100 kΩ to ~1 kΩ, a 100-fold reduction. This allows:
The resistance of the path is 1.67 kΩ. Using 50 times higher resistance would require dangerous voltages (501 kV), cause severe burns from 50 times higher power dissipation, and make defibrillation ineffective.
During open-heart surgery, a defibrillator can be used to bring a patient out of cardiac arrest. The resistance of the path is $500 \text{Ω}$ and a 10.0-mA current is needed. What voltage should be applied?
Strategy
This problem applies Ohm’s Law: $V = IR$. During open-heart surgery, the defibrillator electrodes can be placed directly on the heart, resulting in a much lower resistance path than external defibrillation (which must pass current through the chest wall). We use the given resistance and required current to find the necessary voltage.
Solution
Convert current to SI units and apply Ohm’s Law:
Discussion
The required voltage of 5.00 V is remarkably low compared to the 10,000 V used for external defibrillation (see earlier problem). This dramatic difference occurs because during open-heart surgery, the electrodes are placed directly on the heart muscle, eliminating the high resistance of the chest wall, skin, and other tissues. The direct contact means much less voltage is needed to deliver the therapeutic current. This lower voltage is also much safer for the surgical team and reduces the risk of electrical burns to cardiac tissue. The 10.0 mA current is sufficient to depolarize the heart muscle and restore normal rhythm when applied directly.
A voltage of 5.00 V should be applied to produce the required 10.0-mA current through the 500-Ω path during open-heart surgery.
(a) A defibrillator passes 12.0 A of current through the torso of a person for 0.0100 s. How much charge moves? (b) How many electrons pass through the wires connected to the patient? (See figure two problems earlier.)
Strategy
For part (a), we use the definition of current: $I = Q/t$, where $Q$ is the charge and $t$ is the time. Solving for $Q$ gives us the total charge that flows. For part (b), we use the fact that each electron carries a charge of $e = 1.60 \times 10^{-19}$ C to find the number of electrons.
Solution
(a) From the definition of current:
Solving for charge:
(b) The number of electrons is:
Discussion
Part (a): The charge of 0.120 C is substantial—equivalent to the charge stored in a moderate-sized capacitor. During the brief 10-millisecond pulse, this charge flows through the patient’s torso to depolarize the heart muscle and restore normal rhythm. The short duration is important: it delivers enough charge to be therapeutic without causing tissue damage from prolonged current flow.
Part (b): The number $7.50 \times 10^{17}$ electrons is enormous—750 quadrillion electrons! To put this in perspective:
This calculation illustrates a fundamental principle of electricity: even modest currents involve vast numbers of charge carriers. In metals, the drift velocity of electrons is quite slow (millimeters per second), but the enormous density of mobile electrons means even this slow drift produces significant current.
The current of 12.0 A is quite high for medical applications—defibrillators deliver powerful shocks to overwhelm abnormal electrical activity in the heart. For comparison, as little as 0.1 A across the heart can be fatal under normal circumstances, but during defibrillation, the brief, controlled pulse is therapeutic rather than harmful.
The charge that moves is 0.120 C, corresponding to $7.50 \times 10^{17}$ electrons passing through the wires.
A clock battery wears out after moving 10 000 C of charge through the clock at a rate of 0.500 mA. (a) How long did the clock run? (b) How many electrons per second flowed?
Strategy
For part (a), we use the definition of current $I = \frac{\Delta Q}{\Delta t}$ solved for time: $\Delta t = \frac{\Delta Q}{I}$. For part (b), we convert the current (charge per second) to electrons per second by dividing by the charge of one electron.
Solution
(a) Find the time the clock ran:
Converting to more meaningful units:
(b) Find the number of electrons per second:
Discussion
The clock running for about 7.7 months on a single battery is quite reasonable for a small wall clock or desk clock with a low-power quartz movement. Quartz clocks use very little current because they only need to power the oscillator circuit and step motor. The 3.13 × 10^15 electrons per second sounds enormous, but this is actually a very small current—it takes over 3 quadrillion electrons each second just to produce half a milliamp of current. This illustrates how incredibly small the charge of a single electron is.
(a) The clock ran for $2.00 \times 10^{7}$ s, or about 231 days (approximately 7.7 months). (b) The flow rate was $3.13 \times 10^{15}$ electrons per second.
The batteries of a submerged non-nuclear submarine supply 1000 A at full speed ahead. How long does it take to move Avogadro’s number ( $6.02 \times 10^{23}$ ) of electrons at this rate?
Strategy
First calculate the total charge corresponding to Avogadro’s number of electrons: $\Delta Q = N_A \times e$. Then use $\Delta t = \Delta Q / I$ to find the time.
Solution
Calculate the total charge for $6.02 \times 10^{23}$ electrons:
Calculate the time required:
Discussion
It takes only about 1.5 minutes to move a mole of electrons at this high current! The submarine’s batteries must supply enormous amounts of charge, which is why battery capacity is a critical limitation for non-nuclear submarines. At 1000 A, the submarine is drawing massive power, limiting how long it can operate at full speed before needing to recharge its batteries (typically by surfacing and running diesel generators).
It takes 96.3 s (about 1.6 minutes) to move Avogadro’s number of electrons at 1000 A.
Electron guns are used in X-ray tubes. The electrons are accelerated through a relatively large voltage and directed onto a metal target, producing X-rays. (a) How many electrons per second strike the target if the current is 0.500 mA? (b) What charge strikes the target in 0.750 s?
Strategy
For part (a), we convert the current from charge per second to electrons per second by dividing by the electron charge. For part (b), we use the definition of current rearranged to find charge: $\Delta Q = I \cdot \Delta t$.
Solution
(a) Find the number of electrons per second:
(b) Find the charge striking the target in 0.750 s:
Discussion
The electron gun in an X-ray tube produces over 3 quadrillion electrons per second striking the metal target. When these high-speed electrons suddenly decelerate upon hitting the target, they produce X-rays through bremsstrahlung (“braking radiation”). The 0.500 mA beam current is typical for diagnostic X-ray equipment. The charge of 0.375 mC delivered in 0.750 s is small but represents enormous numbers of individual electrons, each contributing to X-ray production. The intensity of X-rays produced is directly related to this electron current.
(a) Approximately $3.13 \times 10^{15}$ electrons per second strike the target. (b) A charge of 0.375 mC (or $3.75 \times 10^{-4}$ C) strikes the target in 0.750 s.
A large cyclotron directs a beam of ${\text{He}}^{++}$ nuclei onto a target with a beam current of 0.250 mA. (a) How many ${\text{He}}^{++}$ nuclei per second is this? (b) How long does it take for 1.00 C to strike the target? (c) How long before 1.00 mol of ${\text{He}}^{++}$ nuclei strike the target?
Strategy
Each ${\text{He}}^{++}$ nucleus has lost 2 electrons, so it carries a charge of +2e. For part (a), we find the number of nuclei per second from the current and charge per nucleus. For part (b), we use $I = Q/t$. For part (c), we use Avogadro’s number to find the charge in 1 mole of ${\text{He}}^{++}$.
Solution
(a) Each ${\text{He}}^{++}$ nucleus has charge:
Current is $I = 0.250 \text{ mA} = 2.50 \times 10^{-4} \text{ A}$. The number of nuclei per second:
(b) From $I = Q/t$:
(c) One mole contains $N_A = 6.02 \times 10^{23}$ nuclei. Total charge in 1 mole:
Time required:
Discussion
Part (a): The rate of $7.81 \times 10^{14}$ nuclei per second sounds enormous, yet it produces a tiny current of only 0.25 mA. This illustrates that even small currents involve vast numbers of charge carriers.
Part (b): To deliver 1.00 C takes about 67 minutes. This demonstrates that the current is quite small—typical household currents are thousands of times larger.
Part (c): To deliver 1 mole of helium nuclei would take 24.4 years of continuous operation! This calculation shows why particle accelerators rarely accumulate macroscopic amounts of material, despite operating at high particle rates. The total charge of 193,000 C in 1 mole reflects the fact that each ${\text{He}}^{++}$ carries twice the elementary charge.
Cyclotrons are used for nuclear physics research, medical isotope production, and cancer treatment. While the particle rates are high, the actual mass of material delivered is tiny because atomic nuclei are so small. A typical target in a cyclotron experiment might accumulate only micrograms of material even after hours of bombardment.
Answer: (a) $7.81 \times 10^{14}$ nuclei/s, (b) 4.00 × 10³ s (67 min), (c) 7.71 × 10⁸ s (24.4 years)
Repeat the above example on [Example 3], but for a wire made of silver and given there is one free electron per silver atom.
Strategy
We follow the same approach as Example 3: calculate the drift velocity using $I = nqAv_d$, solving for $v_d = \frac{I}{nqA}$. We need the density of silver ($10.49 \times 10^{3}$ kg/m³) and its atomic mass (107.87 g/mol) to find the free electron density $n$. We use the same 12-gauge wire (diameter 2.053 mm) and 20.0-A current as in Example 3.
Solution
First, calculate the free electron density in silver:
The cross-sectional area of the 12-gauge wire is the same as in Example 3:
Now solve for the drift velocity:
Discussion
The drift velocity in silver ($-6.45 \times 10^{-4}$ m/s) is larger in magnitude than in copper ($-4.53 \times 10^{-4}$ m/s from Example 3). This occurs because silver has a lower free electron density than copper ($5.86 \times 10^{28}$ vs. $8.34 \times 10^{28}$ electrons/m³). With fewer charge carriers available, each electron must move faster on average to carry the same current. The negative sign indicates electrons move opposite to the conventional current direction. Despite the faster drift velocity, silver is still an excellent conductor—in fact, silver has the highest electrical conductivity of all metals, though it is more expensive than copper.
The drift velocity of electrons in a 12-gauge silver wire carrying 20.0 A is $-6.45 \times 10^{-4}$ m/s.
Using the results of the above example on [Example 3], find the drift velocity in a copper wire of twice the diameter and carrying 20.0 A.
Strategy
Doubling the diameter quadruples the cross-sectional area (since $A = \pi r^2 \propto d^2$). Use the drift velocity equation $v_d = I/(nqA)$ with the same current and electron density as Example 3.
Solution
The original wire has diameter 2.053 mm, so the new diameter is:
The new cross-sectional area:
This is 4 times the original area ($4 \times 3.310 \times 10^{-6} = 1.324 \times 10^{-5}$ m²).
Using the electron density from Example 3 ($n = 8.342 \times 10^{28}$ electrons/m³ for copper):
Discussion
The drift velocity is exactly 1/4 of the original value ($-4.53 \times 10^{-4}$ m/s ÷ 4 = $-1.13 \times 10^{-4}$ m/s), as expected since quadrupling the area while keeping current constant reduces drift velocity proportionally. This demonstrates why larger wires can safely carry higher currents—the electrons move more slowly, reducing collisions and heat generation per unit volume.
The drift velocity in the larger copper wire is $-1.13 \times 10^{-4}$ m/s (about 0.113 mm/s).
A 14-gauge copper wire has a diameter of 1.628 mm. What magnitude current flows when the drift velocity is 1.00 mm/s? (See above example on [Example 3] for useful information.)
Strategy
We use the current-drift velocity relationship $I = nqAv_d$ directly. From Example 3, we know the free electron density in copper is $n = 8.342 \times 10^{28}$ electrons/m³. We need to calculate the cross-sectional area of the 14-gauge wire and convert the drift velocity to SI units.
Solution
First, calculate the cross-sectional area of the 14-gauge wire:
Convert the drift velocity to SI units:
Now calculate the current (using the magnitude of charge):
Discussion
A current of 27.8 A is quite large for a 14-gauge wire. In fact, 14-gauge wire is typically rated for only about 15-20 A in household wiring applications because of heating concerns. The drift velocity of 1.00 mm/s is also relatively high compared to typical values (which are on the order of 0.1 mm/s or less for household currents). This problem illustrates that even modest increases in drift velocity result in substantial currents because of the enormous number of free electrons available in metals. It also shows why electrical codes limit currents in specific wire gauges—excessive current causes heating that can damage insulation or cause fires.
A current of 27.8 A flows through the 14-gauge copper wire when the drift velocity is 1.00 mm/s.
SPEAR, a storage ring about 72.0 m in diameter at the Stanford Linear Accelerator (closed in 2009), has a 20.0-A circulating beam of electrons that are moving at nearly the speed of light. (See [Figure 8].) How many electrons are in the beam?
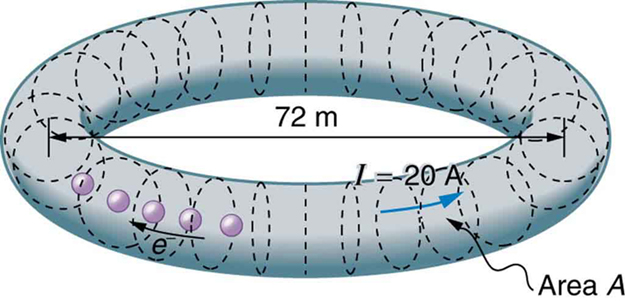
Strategy
The electrons complete the circular path at nearly the speed of light. Current is related to the charge passing a point per unit time. The time for one orbit is $T = \text{circumference}/v = \pi d/c$. The total charge in the beam is $Q = IT$, and dividing by the electron charge gives the number of electrons.
Solution
Calculate the circumference of the storage ring:
Calculate the time for one orbit at nearly the speed of light ($c = 3.00 \times 10^{8}$ m/s):
The total charge in the beam equals current times orbital period:
The number of electrons:
Discussion
The beam contains about 94 trillion electrons—a large number, but still only about $1.6 \times 10^{-10}$ moles, far too small to weigh. Despite the high current of 20 A, each electron passes any given point many times per second (about 1.3 million orbits per second), so relatively few electrons are needed to maintain this current. The electrons travel at nearly the speed of light, making many complete circuits each second, which is why a modest number of particles can produce such a large current.
The SPEAR storage ring contains approximately $9.42 \times 10^{13}$ electrons.